
qwertyuiopasdfghjklzxcvbnmqw
ertyuiopasdfghjklzxcvbnmqwert
yuiopasdfghjklzxcvbnmqwertyui
opasdfghjklzxcvbnmqwertyuiopa
sdfghjklzxcvbnmqwertyuiopasdf
ghjklzxcvbnmqwertyuiopasdfghj
klzxcvbnmqwertyuiopasdfghjklz
xcvbnmqwertyuiopasdfghjklzxcv
bnmqwertyuiopasdfghjklzxcvbn
mqwertyuiopasdfghjklzxcvbnmq
wertyuiopasdfghjklzxcvbnmqwe
rtyuiopasdfghjklzxcvbnmqwerty
uiopasdfghjklzxcvbnmqwertyuio
pasdfghjklzxcvbnmqwertyuiopas
dfghjklzxcvbnmqwertyuiopasdfg
hjklzxcvbnmqwertyuiopasdfghjk
lzxcvbnmrtyuiopasdfghjklzxcvbn
Chemical Composition of Teeth
DENTAL CHEMISTRY
2012
Prof.Dr. Amer Taqa

Chemical Composition of Teeth
Sample preparation
Mechanical methods
A diamond saw or cutting disc is usually used to obtain layers of enamel or dentine or thin
sections of longitudinal or horizontal planes for use as slides in light microscopy or electron
probe analysis.
Large amounts of dentine or enamel material are usually prepared by using a diamond griding
wheel with great care to avoid contaminating thr desired material.
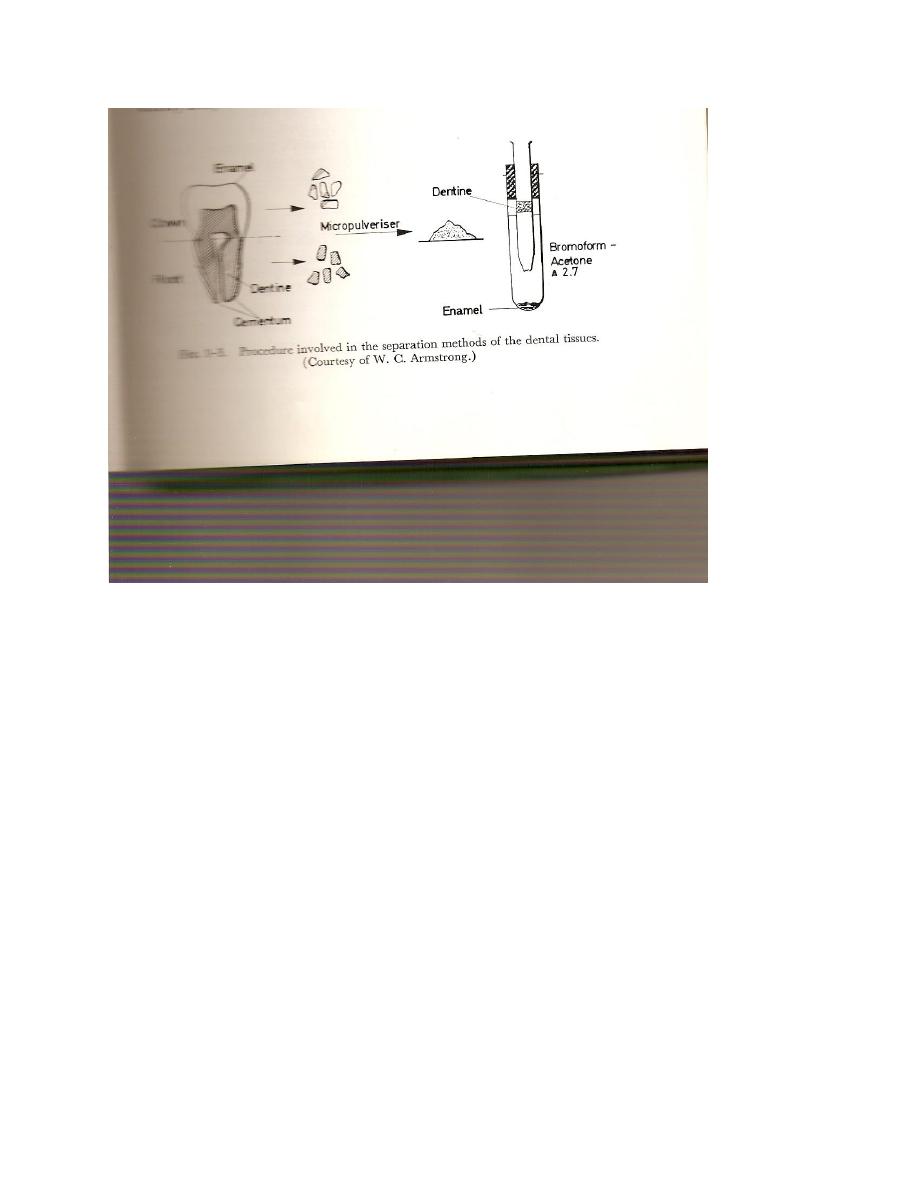
Flotation technique of Manly and Hodge
Once the tooth has been pulverized , the more dense enamel particles can be separated from the
dentine by using a flotation or centerfuge-flotation technique. The particles are interoduced into
a solution of acetone –bromo – from having a specific gravity of 2.70. enamel having a density
of 2.92±0.1 settles during centrifugation or flotation, while the lighter dentine with a density of
about 2.40
floats on the surface permitting either fraction to be readily collected(see figure). A single
separation gives enamel and dentine of 99% purity as determined by a refractive index method.
Cementum, density 2/03, can also be separated by this technique.
Chemical methods
Acids have been used to etch away successive layers of enamel and dentine to give either
solutions of the desired soluble material or pure residues of wanted material. By adjusting the
strength of the acid and the etching time, any thickness from as low as 10 microns may be
achieved.
Chemical composition
A tooth unlike a shaker full of recrystalized salts, has no single, constant, chemical
stoichiometry. It has been constituted and shaped by unique genetic and biochemical individual
and therefore can be varied as nature will permit.
One reporting the composition of a tooth one must constantly keep in mind the effects of diet,
position in the mouth , geographical locality, age, condition of the tooth and medical history of

the contributing individual. In the instances where these facts and their effects are known, they
will be noted.
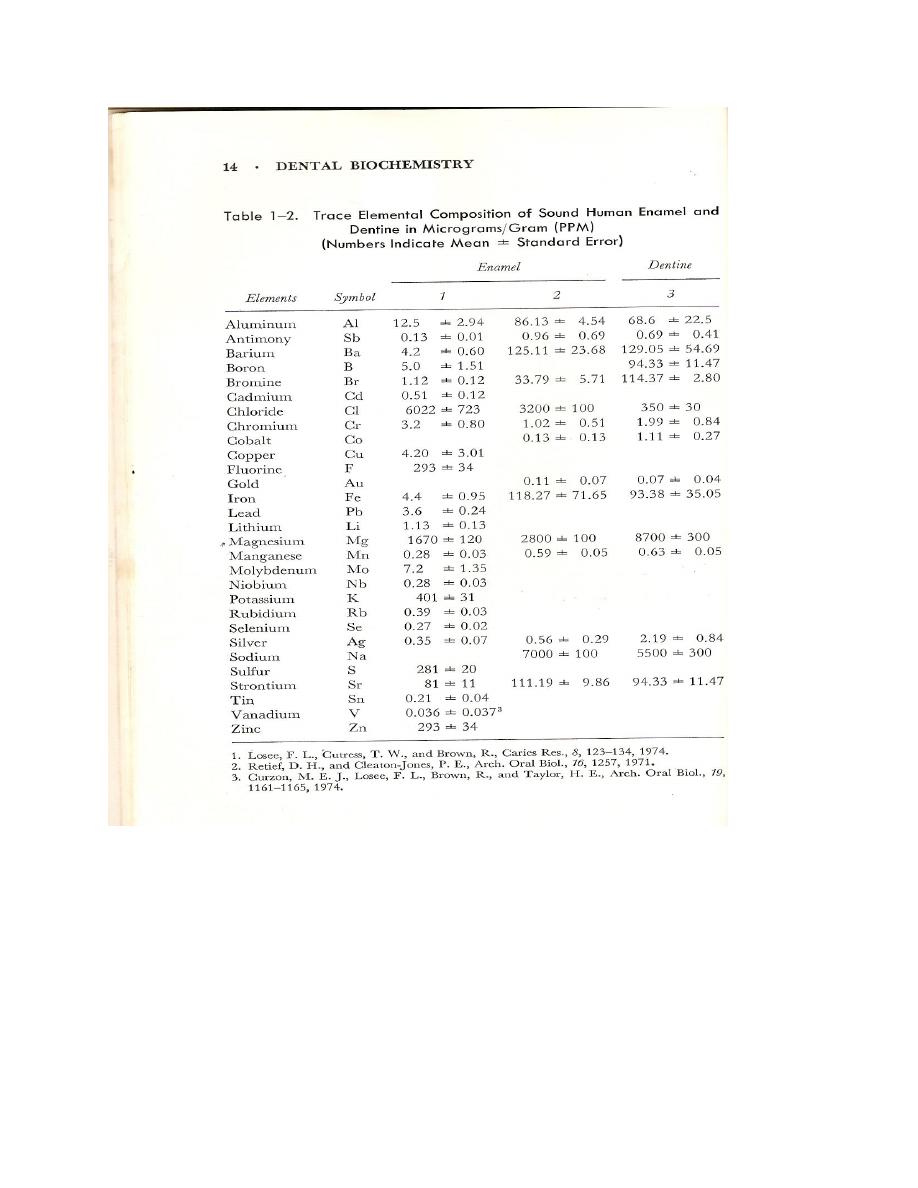
Inorganic constituents
Tooth analysis
In 1973 Lefever and Hodge reported the values intable for the chemical analysis of teeth. Their
data permitted the following conclusions:

1- Deciduous teeth have more moister, less inorganic residue , Ca and P, and about the
same carbonate content as permanent.
2- There is little differences, except in moister content, between sound and carious teeth.
3- Age causes no change in the chemical constitution of teeth.
4- There is little chemical difference between teeth from male and female patients
5- Increasing severity of pyorrhea may cause a decrease in the carbonate content of teeth.
6- The composition of teeth substance is remarkably constant.
Calcium and phosphorus
Ca/P retio are slightly lower in carious than in sound enamel(table). Sound enamel from age
groups beyond 30 years has a lower Ca/P ratio(1.97) than sound enamel from the younger age
group(2.07).
As can be ssen from the table thr Ca/P ratio of enamel and dentines lies between that of
octacalcium phosphate , Ca
8
H
2
(PO
4
)+5H
2
O, 1.72 and hydroxyapatite, Ca
10
(PO
4
)
6
(OH)
2
,2.15.
possible intermidate compounds include hydrated tricalcium phosphate . two theories have been
formulated to explain the continuous series of apatitic calcium phosphate.
1- The absorption theory, in which acid phosphate groups absorb to microcrystalline
hydroxyapatite.
2- The defect theory, which proposes that hydrogen ions in hydroxapatite are substituted for
Ca ions.
Water:
Enamel humidified to 100% relative humidity loses 1.7% and 2.1% by weigh of water at 61 and
100C, respectively. The use of nuclear magnetic resonance (NMR) revealed that heating to 200C
was insufficient to dehydrate dental enamel.
Carbohydrate
Carbon dioxide (carbonate), unlike zinc, lead or fluoride , has a reverse distribution pattern. An
ealier study gave the content of the outer enamel surface to be about 1.5% by weight increasing
in smooth (concave) curve to about 2.9% by weight that the dentino-enamel junction (DEJ).
Magnesium

Brudevold and his co-workers showed that that the surface enamel has a lower Mg content than
the body on intact enamel , 30-60 versus 60-74mM per gm.
Fluoride
Most investigators agree that the caries –inhibiting effect of fluoride is due to its relatively high
concentration in the surface layer of the enamel .
The continuous drinking of water containing 0.1-0.5 ppm of fluoride by person under 20 years
of age caused the level of surface enamel fluoride to rise from 419 to 3,370ppm. A degree of
caries protections occurs when one hydroxyl group of hydroxyapatite ions is replaced by a
fluoride per surface unit cell.
The inhibition of caries found in fluoride areas is due to the presence of fluoride alone and not to
change in other enamel components.
Fluoride concentration in deciduous teeth follows a pattern similar to that in permanent teeth,
although the level in the enamel surface to approximately 30 microns in depth are lower than in
the permanent teeth.
Chloride
Chloride is capable of exchanging with the hydroxyl group of hydroxyapatite but is not fixed in
calcified tissues.
Strontium
The uptake of strontium occurs prior to eruption , probably during tooth formation , since their is
no change in concentration with age. The level of strontium concentration is about constant in
the surface and subsurface enamel.
Vanadium
Vanadium was not considered as one of the essential trace elements but, after showing that is
necessary for the growing rat. In 1972 it was added to the list of elements essential for life.
Lead
Deciduous teeth have been found to give an excellent indication of the lead accumulation in
individual children.
Studies on whole teeth indicate that

1. The amount of lead increase at a fairly uniform rate through age 50(11 to27ppm), when a
rapid increase occurs that levels off (50-55ppm) until age 79.
2. There is little different in assimilation by males and females.
3. Urban residents had lower levels than those of sub-urban or rural residents.
4. Ancient teeth (A.D. 200-600) had low metal level and no age correlation (5.3 to 2.9ppm).
5. Cadmium (3.1 to 5.8 ppm0 and zinc (1.6 to 3.6 ppm) levels showed no correlation with
age.
Trace elements
Trace elements can be divided into three categories:
1. Those which apper to have no biological role and which are present in tissue only as
adventitious contamination from the environment.

2. Those elements which appear to be essential to the enzymatic processes of living cell( eg.
Fe,Zn,Cu,I,Mo,I,Co,Mn,Se).
3. Elements which are probably essential nutrients but whose metabolic action in not clear
(e,g. F,Br,Ba,Sr).
Trace elements may assist in reducing caries by altering tooth solubility , by changing tooth
morphology , or by altering the size and or shape of the crystallites and ultimately, the enamel
structure.
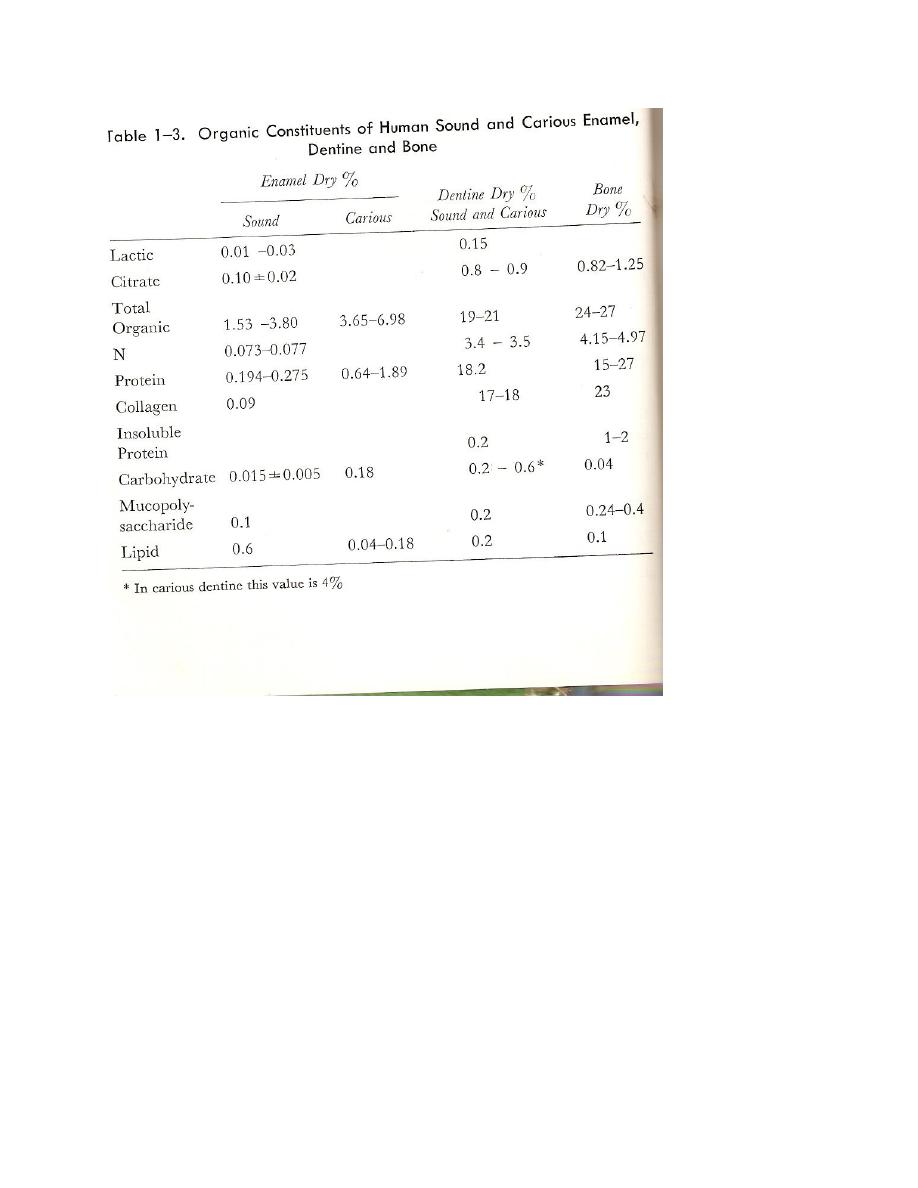
Organic constituents
Citrate
Citrate occurs in greater concentration in the surface and junction enamel than in the body of the
enamel going from value 3.5mM/gm to 1.1 and back to about 4.4mM/gm. Whether the
distribution varies with age has not yet been determined.
Citrate , which has been found in all mineralized tissues, may be
1- An accidental co precipitation component of calcium phosphates.
2- In a citrate- containing arginine-rich peptide .
3- In the form of phosphate or pyrophosphoric-citrate.
Lactate
Lactate follows almost the same distribution and content as citrate and it is possible that both are
located primarily in the water in the enamel since a comparison shows similar curves.
Nitrogen
The amount of nitrogen can be used as a measure of the concentration of organic material in
areas of the tooth. The scientists found that there is no change with the age in the N
concentration in enamel except that occurring in the last decade of life when measured on a
weight basies. There is no theoretical basis to except that the organic material in a tooth should
change with age. Possible alter ations may occur from external source through cracks and voids
which would be more numerous in the order tooth due to wear and tear. Teeth over 50 years old
differ from younger teeth by having:
1- Greater N concentration in the surface enamel , 0.15% versus 0.1%.
2- Greater N concentration at the dentino enamel junction 0.2% versus ca. 0.12%
3- Lower N concentration in the body of the enamel for a greater depth, ca.0.04%versus
ca.0.07%N.

Protein in teeth
In discussing the enamel proteins, attention must ber given to the age of the tooth because of
major differences observed between the developing (immature) and the mature tooth. These
differences include
1-total protein content.
2-solubility
3-aminiacid composition
The total protein content of human enamel diminishes from approximately 15 to 20% in the
developing tooth to about 0.05 to 0.2% at maturity. A similar large decrease in the enamel
protein content of the maturing bovine tooth has been observed. An absolute loss of 90% in the
weight of enamel protein during maturation has been demonstrated. The process responsible for
the loss is unknown.

COLLAGEN
Collogen is the major protein of the extracellular connective tissues and functions as a structural
protein serving principally as the prime mechanical support of tissues. The amount of collagen
varies from one species to another and from tissue to tissue within the same species. Collagen
content of normal human dentine has been estimated to be 18% by dry weight.

Physicochemical properties
From the standpoint of protein structure, collagen is an unique molecules.
Structure
In the native state, most collagen is insoluble. However , native soluble collagen can be obtained
in vitro under certain conditions.
A collogen monomer consists of three polypeptide chains, each chain being twisted into a left-
handed helix. The three helices in turn from a right-hunderd helix. The three helixes in turn from
a right –handed super or major helix.
Biosynthesis of collagen
Collagen is formed by several steps:
Hydroxylation of proline and lysine
Helix formation
Glycosylation
Transport and secretion
’procollagen-collagen conversion
Fiber formation
cross-linking

CARBIHYDRATE OF TEETH
Various attempts have been made to extract and characterize the carbohydrate- containing
components of teeth.
It was found that the dentine-cementum and enamel fractions of human teeth contain
respectively 0.08 and 0.03% total aminosugar. Of these amounts glucose amine represents 42
and 47% of the total, respectively in dentine –cementum also contained a third compound
(accounting for 15% of total hexamine).
In some tissues, the synthesis of glycosamine requires not glutamine but ammonia, as indicated
by fallowing reaction;
NH
3
+fructose-6- phosphate ↔ glycosamine-6- phosphate +H
2
O

The glycoamineglycons are high molecular weight hetropolysaccaride they may be conveniently
represented by a ; (repeating unit) which is repeating many times to produce chains of different
length eight different GAG( (glycosaminoglycans) have been isolated and characterized on the
basis of
1- The structure of repeating unit.
2- Optical rotation
3- Solubility in alcoholic solutions
4- Enzymatic degradation
The acidic character of these compounds is due to the presence of three different functional
group
Carboxylin (-COOH)
Ester sulfhate(-O-SO
4
H)
Sulfonamide(-N-SO
3
H)
LIPIDS IN TEETH AND MEMBERANCE

The presence of phosphatidyl serine was confirmed in carious dentin. Since this lipid was not
wextracted from non carious material, a possible binding of inorganic salt by lipid was
suggested, which prompted lipid analysis of deter mineralized dentin matrix. Lipid extracts of
sound and EDTA- dermineralized dentin were separated and quantitated, the results of which are
shown in table:
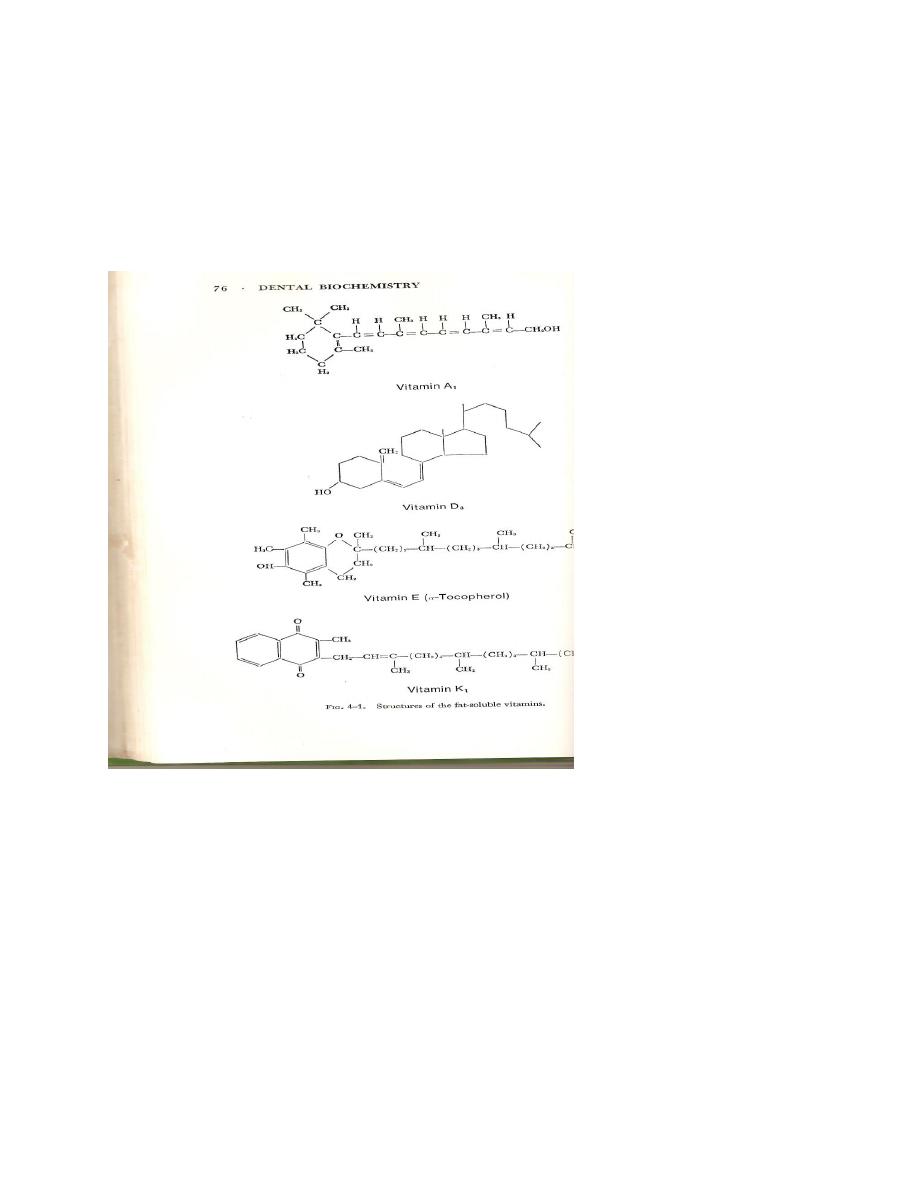
Fat soluble vitamins
Vitamins A,D,E, and K are found associated with the lipid fraction of foods and their absorption
into the body depend upon the presence of bile for emulsification. The structure of the fat soluble
vitamins are shown in figure.
Lipids in membranes
Structure
Basically the phospholipids are the molecules that define membrane structure. These
compounds are amphipathic in nature, which means that they contain both polar and polar
regions.

