
Cardiac arrhythmia

A cardiac arrhythmia is defined as a disturbance of the electrical
rhythm of the heart. Cardiac arrhythmias are often a manifestation of
structural heart disease but may also occur because of abnormal
conduction or depolarisation in an otherwise healthy heart.

Pathogenesis
There are three main mechanisms of tachycardia:
1. Increased automaticity. The tachycardia is produced by
spontaneous depolarisation of an ectopic focus in the atria,
atrioventricular junction or ventricles, often in response to
catecholamines. Single depolarisations lead to atrial, junctional or
ventricular premature (ectopic) beats. Repeated depolarisation leads
to atrial, junctional or ventricular tachycardia.
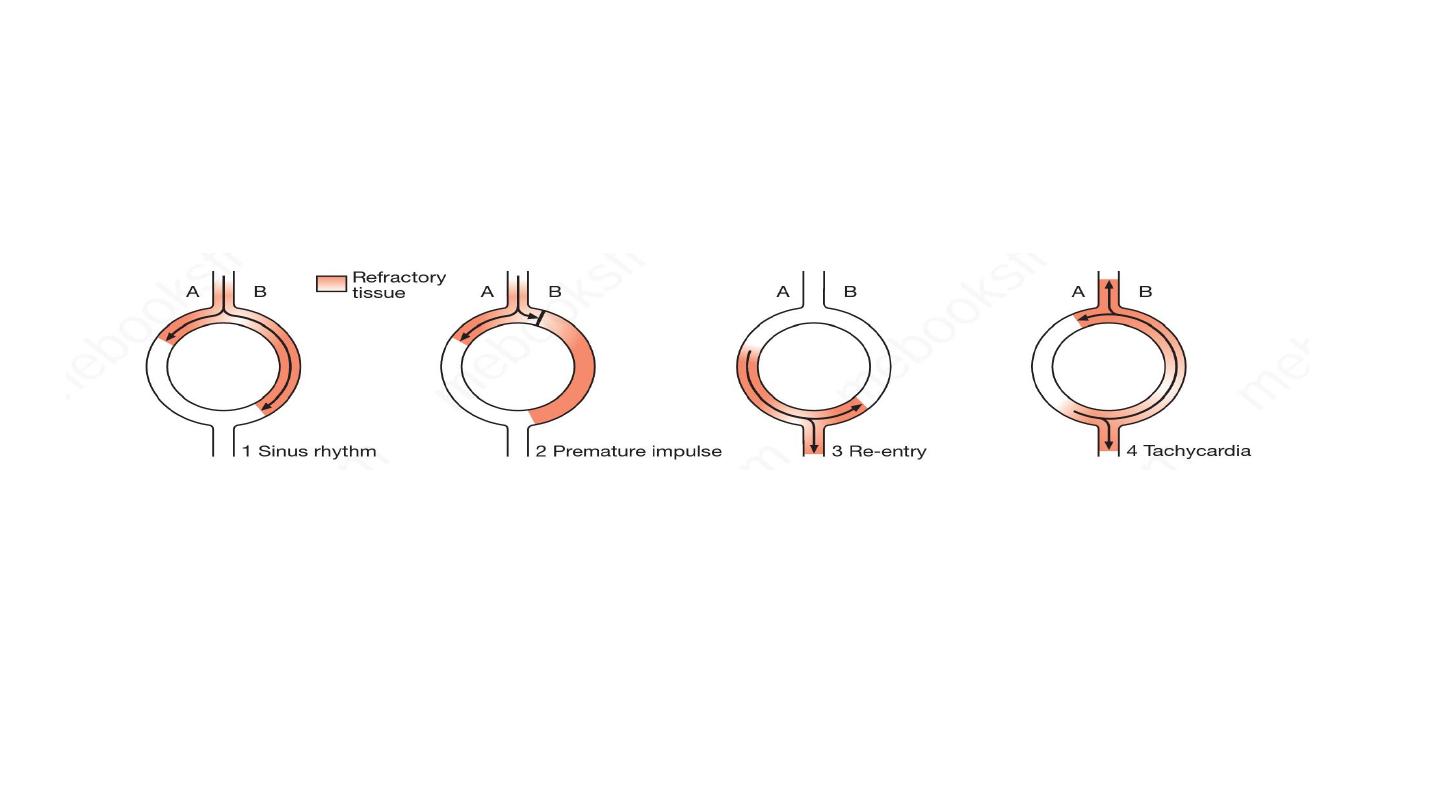
2. Re-entry. The tachycardia is initiated by an ectopic beat and
sustained by a re-entry circuit (Fig. 16.31). Most tachyarrhythmias
are caused by re-entry.
3. Triggered activity. This can cause ventricular arrhythmias in
patients with coronary artery disease. It is a form of secondary
depolarisation arising from an incompletely repolarised cell
membrane. Arrhythmias may be supraventricular (sinus, atrial ,
junctional) or ventricular.

Supraventricular rhythms usually produce narrow QRS complexes
because the ventricles are depolarised in their normal sequence via the
AV node, the bundle of His and associated Purkinje fibres.
In contrast, ventricular rhythms produce broad, bizarre QRS complexes
because the ventricles are activated in an abnormal sequence.
Occasionally, supraventricular tachycardia can mimic ventricular
tachycardia and present as a broad-complex tachycardia due to
coexisting bundle branch block or the presence of an additional
atrioventricular connection (accessory pathway



If the sinus rate becomes unduly slow, another, more distal part
of the conducting system may assume the role of pacemaker. This
is known as an escape rhythm and may arise in the AV node or
His bundle (junctional rhythm) or in the ventricles (idioventricular
rhythm).

Clinical features
Asymptomatic, sustained tachycardia might cause
Palpitation, Dizziness, Chest discomfort, Breathlessness, Syncope
Bradycardias tend to cause symptoms that reflect low cardiac
output, including fatigue, lightheadedness and syncope. Extreme
bradycardias or tachycardias can precipitate sudden death or
cardiac arrest

Investigation
Standard 12 leads ECG
Ambulatory ECG
Pateint activated loop recorder

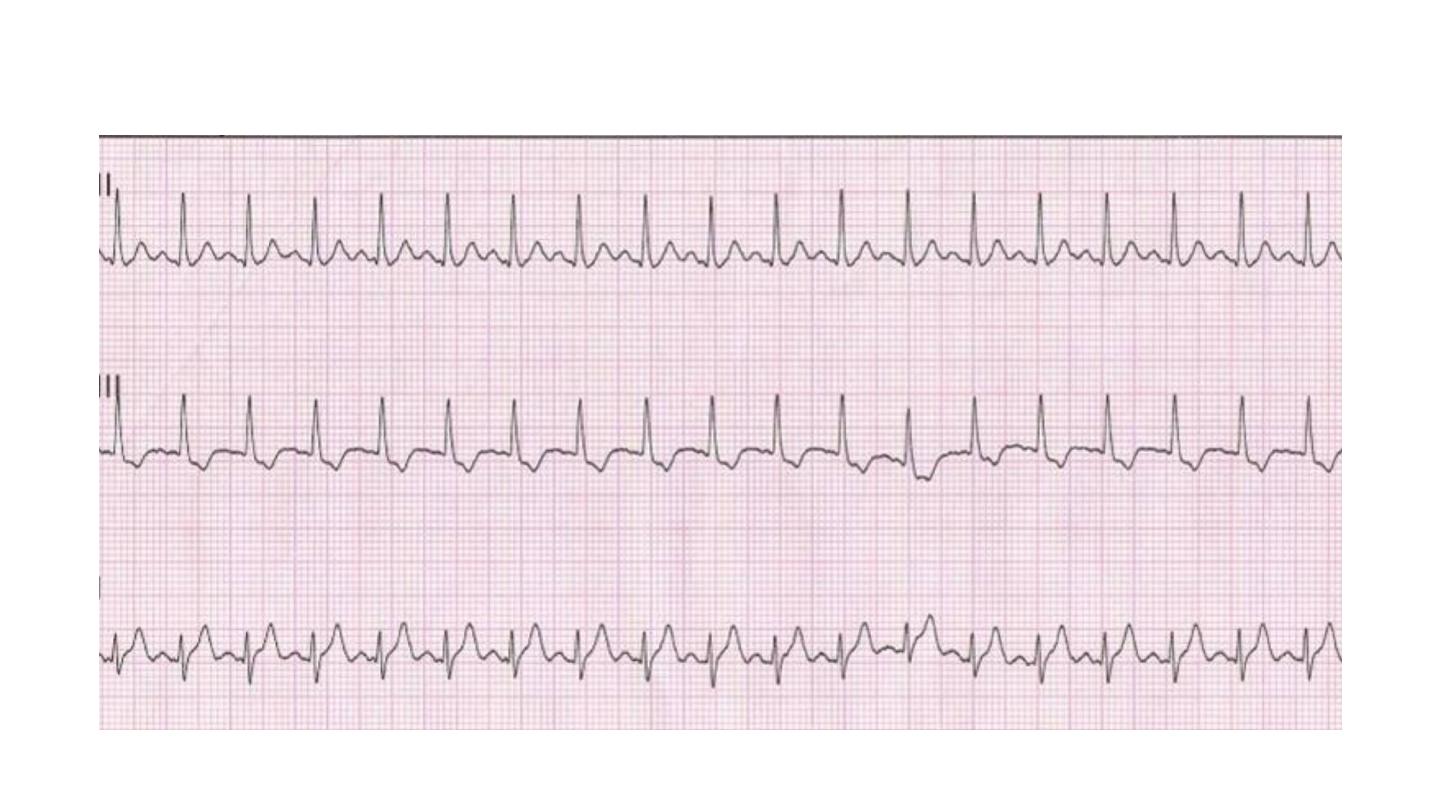
Sinus arrhythmia
defined as a cyclical alteration of the heart rate during respiration, with
an increase during inspiration and a decrease during expiration. Sinus
arrhythmia is a normal phenomenon and can be quite pronounced in
children.
Absence of this normal variation in heart rate with breathing or with
changes in posture may be a feature of diabetic neuropathy, autonomic
involvement in patients with diseases of peripheral nerves or increase
sympathetic drive.
Need no treatment.
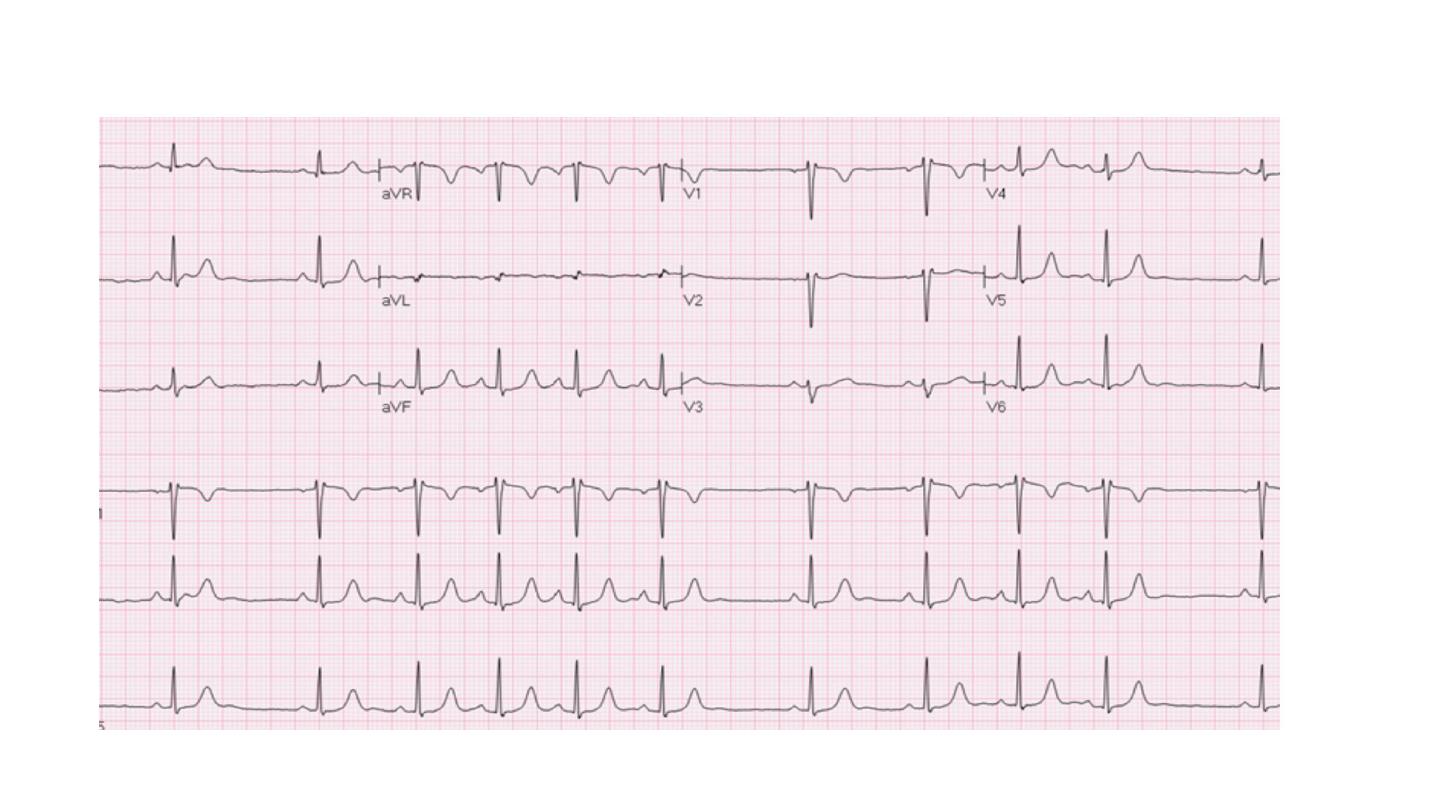

Sinus bradycardia
HR less than 60 beat per minute. May occur in normal people, a
common finding in athlets.
Asymptomatic bradycardia need no treatment
Treatment: atropine , pacing ( temporary or perminant)
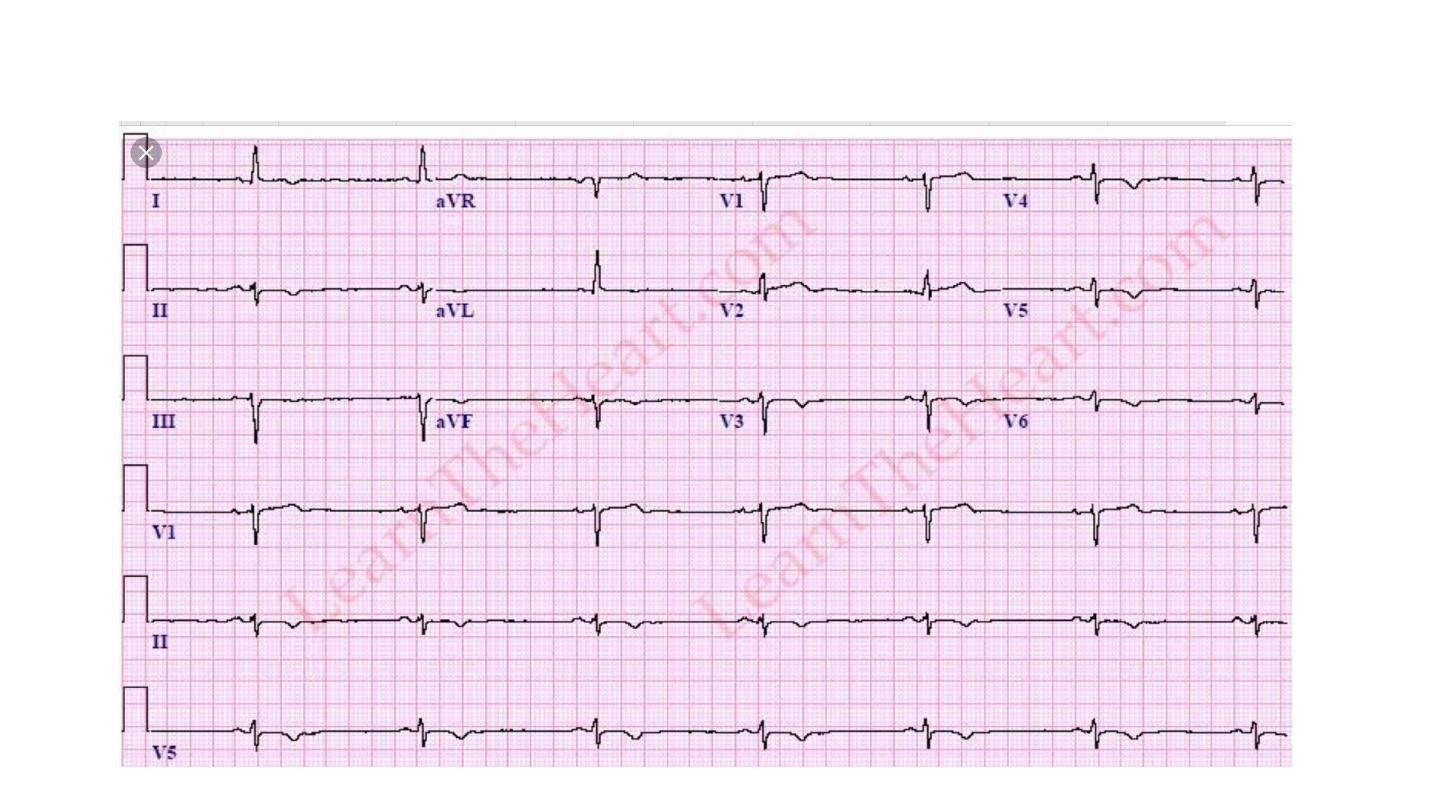

Sinus tachycardia
Sinus tachycardia is usually due to an increase in sympathetic activity
associated with exercise, emotion and pregnancy. Healthy young adults
can produce a rapid sinus rate, up to 200/min, during intense exercise.
Sinus tachycardia does not require treatment but sometimes may
reflect an underlying disease.


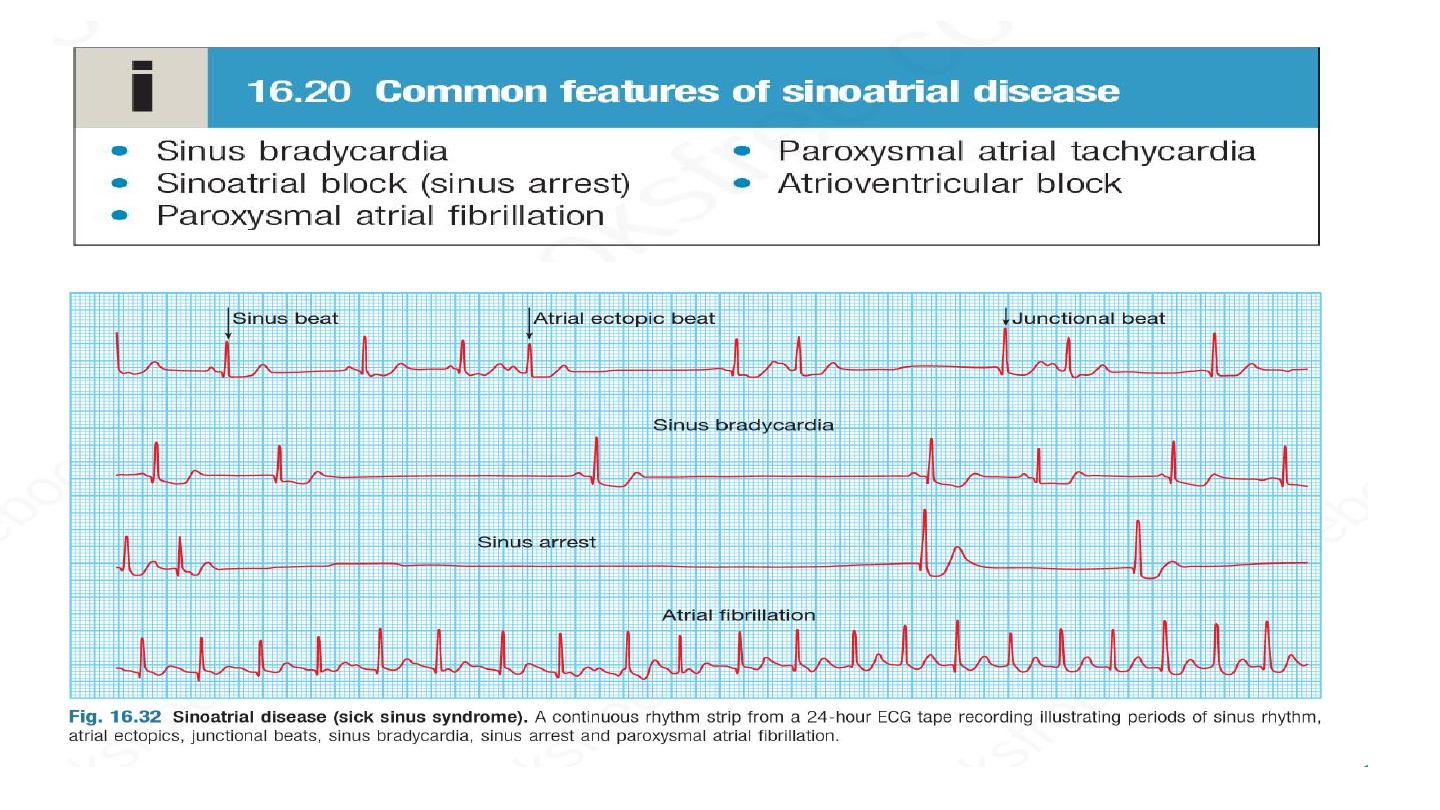
Sinus node dysfunction
Caused by ischemia, fibrosis and degenerative changes of SA node.
Typical presentation is with palpitation, dizzy spells or syncope, due to
intermittent tachycardia, bradycardia, or pauses with no atrial or
ventricular activity (SA block or sinus arrest)
A permanent pacemaker may benefit patients with troublesome
symptoms due to spontaneous bradycardias, or those with
symptomatic bradycardias induced by drug required to prevent
tachyarrhythmias. Atrial pacing may prevent episodes of atrial
fibrillation. Pacing improves symptoms but not prognosis, and is not
indicated in patients who are asymptomatic.

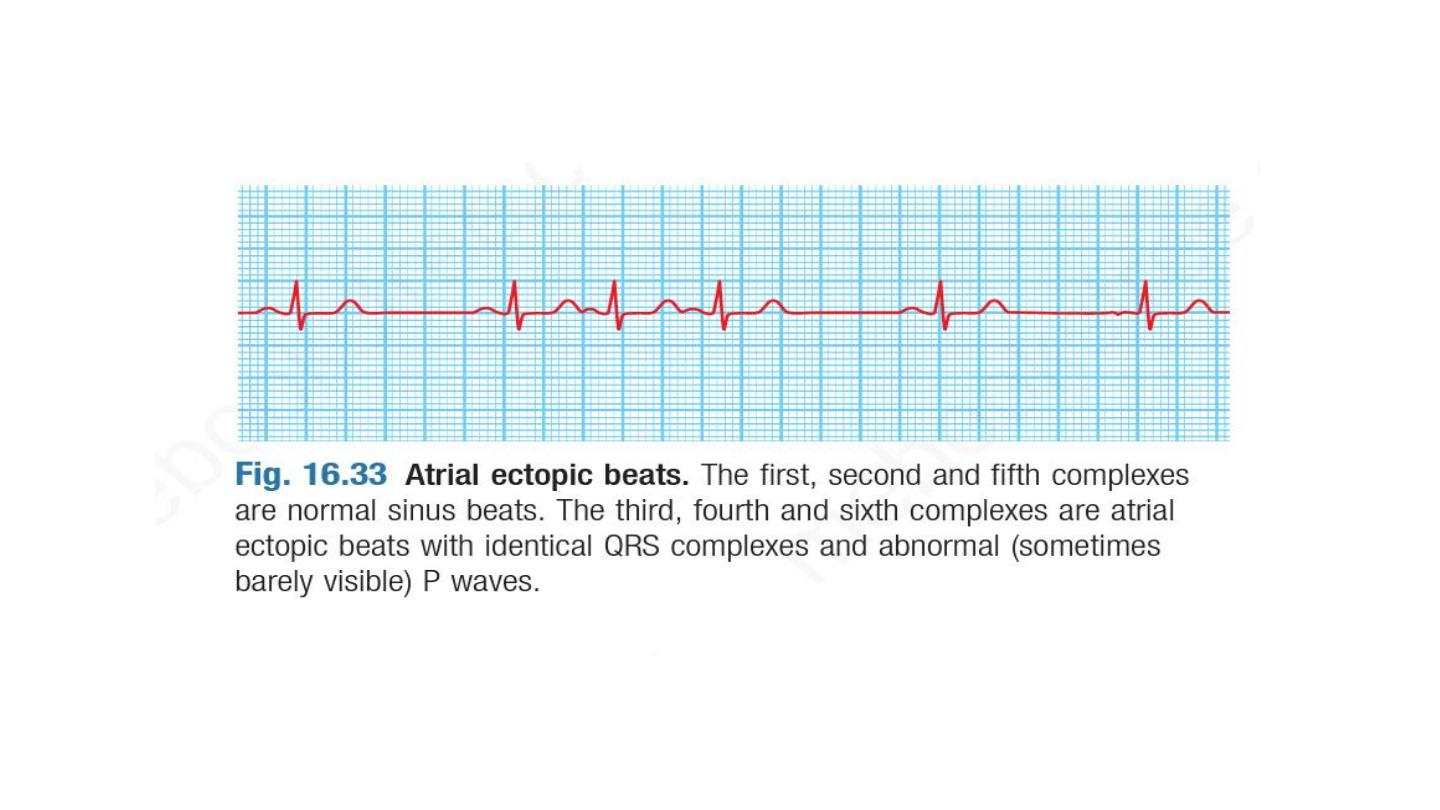
Premature atrial beat ( atrial ectopic)
Usually asymptomatic, or may give sensation of missed beat or
abnormally strong beat
ECG show premature but otherwise normal QRS complex , p wave if
visible has abnormal morphology because the atria activate from
abnormal site.
Usually need no treatment, however frequent atrial e topics may
herald the onset of atrial fibrillation
Beta blocker can be used if symptoms is significant.

Atrial fibrillation
Atrial fibrillation (AF) is the most common sustained cardiac
arrhythmia, with an overall prevalence of 0.5% in the adult
population of the UK.
The prevalence rises with age, affecting 1% of those aged 60–64
years, increasing to 9% of those aged over 80 years. It is
associated with significant morbidity and a twofold increase in
mortality. This is mainly because of its association with underlying
heart disease but also because of its association with systemic
embolism and stroke

Classification
Paraxysmal: intermittent episode of AF that self terminated in 7 days
Persistent: prolong episode that can be terminated by electrical or
pharmacological cardioversion.
Perminant.
Other classifcation is:
valvular AF in prosthetc valves, mitral stenosis, mitral annuloplasty surgery.
Non valvular , other causes
Valvular AF has 10 fold risk of thromboembolism than non valvular AF

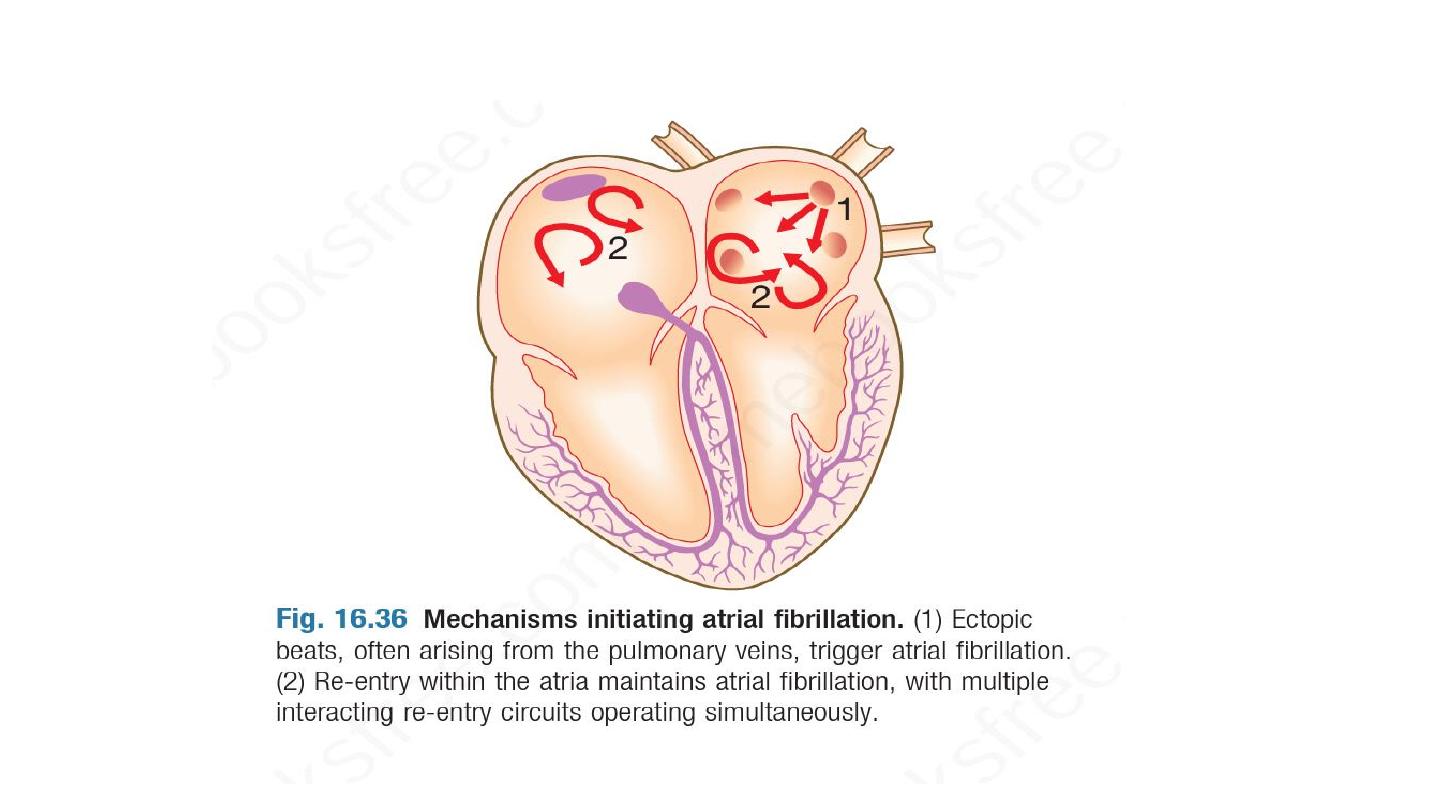
Pathogenesis
AF is a complex arrhythmia characterised by both abnormal
automatic firing and the presence of multiple interacting re-entry
circuits looping around the atria. Episodes of AF are initiated by
rapid bursts of ectopic beats arising from conducting tissue in the
pulmonary veins or from diseased atrial tissue. It becomes
sustained because of re-entrant conduction within the atria or
sometimes because of continuous ectopic firing.
LA size correlate with the risk of AF
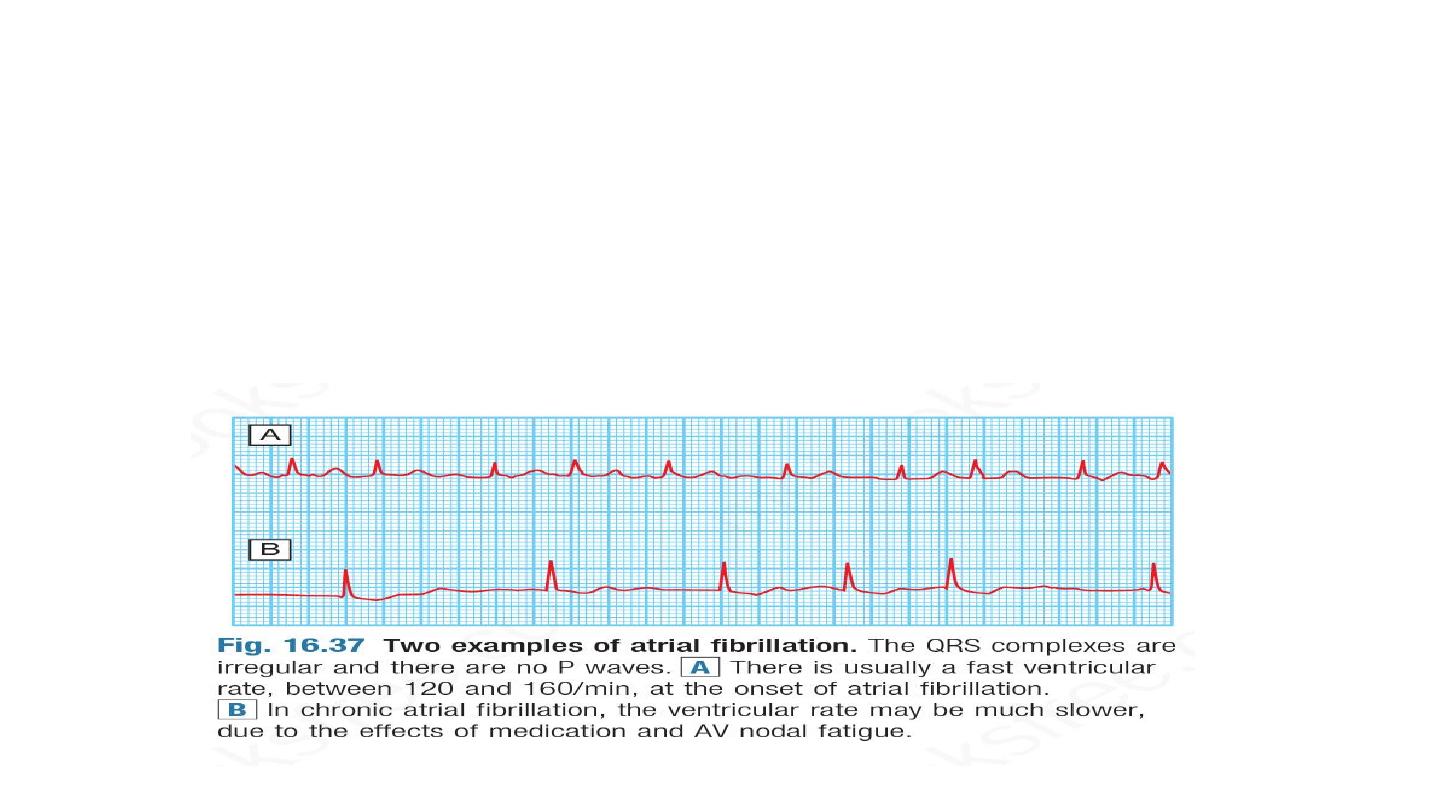
During episodes of AF, the atria beat rapidly but in an
uncoordinated and ineffective manner. The ventricles are activated
irregularly at a rate determined by conduction through the AV
node. This produces the characteristic ‘irregularly irregular’ pulse.
The ECG shows normal but irregular QRS complexes; there are no
P waves but the baseline may show irregular f ibrillation waves.


Alcohol excess, hyperthyroidism and chronic lung disease are also
common causes of AF, although multiple predisposing factors may
coexist, such as the combination of alcohol, hypertension and
coronary artery disease.
About 50% of all patients with paroxysmal AF and 20% of patients
with persistent or permanent AF have structurally normal hearts;
this is known as ‘
lone
atrial fibrillation’.

Clinical features
The typical presentation is with palpitation, breathlessness and
fatigue.
In patients with poor ventricular function or valve disease, AF may
precipitate or aggravate cardiac failure due to loss of atrial
contraction and the rapid ventricular response.
In elderly AF may be asymptomatic and discovered incidently.

Investigation
12 lead ECG
Echocardiography
TFT
TMT & coronary angio if IHD is a possible cause

Managment
Rate control
Rhythm control
Prevention of recurrence
Prevention of thromboembolism

Rate control
Beta blocker
Calcium channel blocker
Digoxin
AV node ablation and perminant pacemaker implantation ( pace and
ablate strategy)
Beta-blockers and rate-limiting calcium antagonists are more
effective than digoxin at controlling the heart rate during exercise
and have additional benefits in patients with hypertension or
structural heart disease

Rhythm control
Cardioversion is initially successful in most patients but relapse
is frequent (25–50% at 1 month and 70–90% at 1 year).
Attempts to restore and maintain sinus rhythm are most
successful if AF has been present for less than 3 months, the
patient is young and there is no important structural heart
disease.
Cardioversion is either pharmacological or electrical.
Electrical cardioversion if there is hemodynamic instability or no
response to pharmacological cardioversion

If onset of AF less than 48 hours, cardioversion can be given and intravenous
heparin should be given
If onset is unknown or more than 48 hours, TEE should be done to R/O LAA
thrombus, or anticoagulation with warfarin or NOAC for 4 weeks should be given
before cardioversion.
In stable patients with no history of structural heart disease, intravenous
flecainide (2 mg/kg over 30 mins, maximum dose 150 mg) can be used for
pharmacological cardioversion and will restore sinus rhythm in 75% of
patients within 8 hours.
In patients with structural heart disease intravenous amiodarone can be given.
Pills in pocket regimen: flecainide 200 to 300 mg orally can be used to restore
sinus rhythm in young patient without structural heart disease if proved to be
safe and effective in hosoital to restore sinus rhythm.

AF catheter ablation
Pulmonary vein isolation

Prevention of recurrence
Class Ic drugs such as propafenone or flecainide, are also effective at
preventing episodes but should not be given to patients with coronary
artery disease or left ventricular dysfunction.
Flecainide is seldom used alone, since it can precipitate atrial flutter, and is
usually prescribed with a rate-limiting β-blocker.
Class III drugs can also be used; amiodarone is the most effective agent for
preventing AF but side-effects restrict its use to when other measures fail.
Dronedarone is an effective alternative but is contraindicated in patients
with heart failure or significant left ventricular impairment.

Prevention of thrmboemblosim
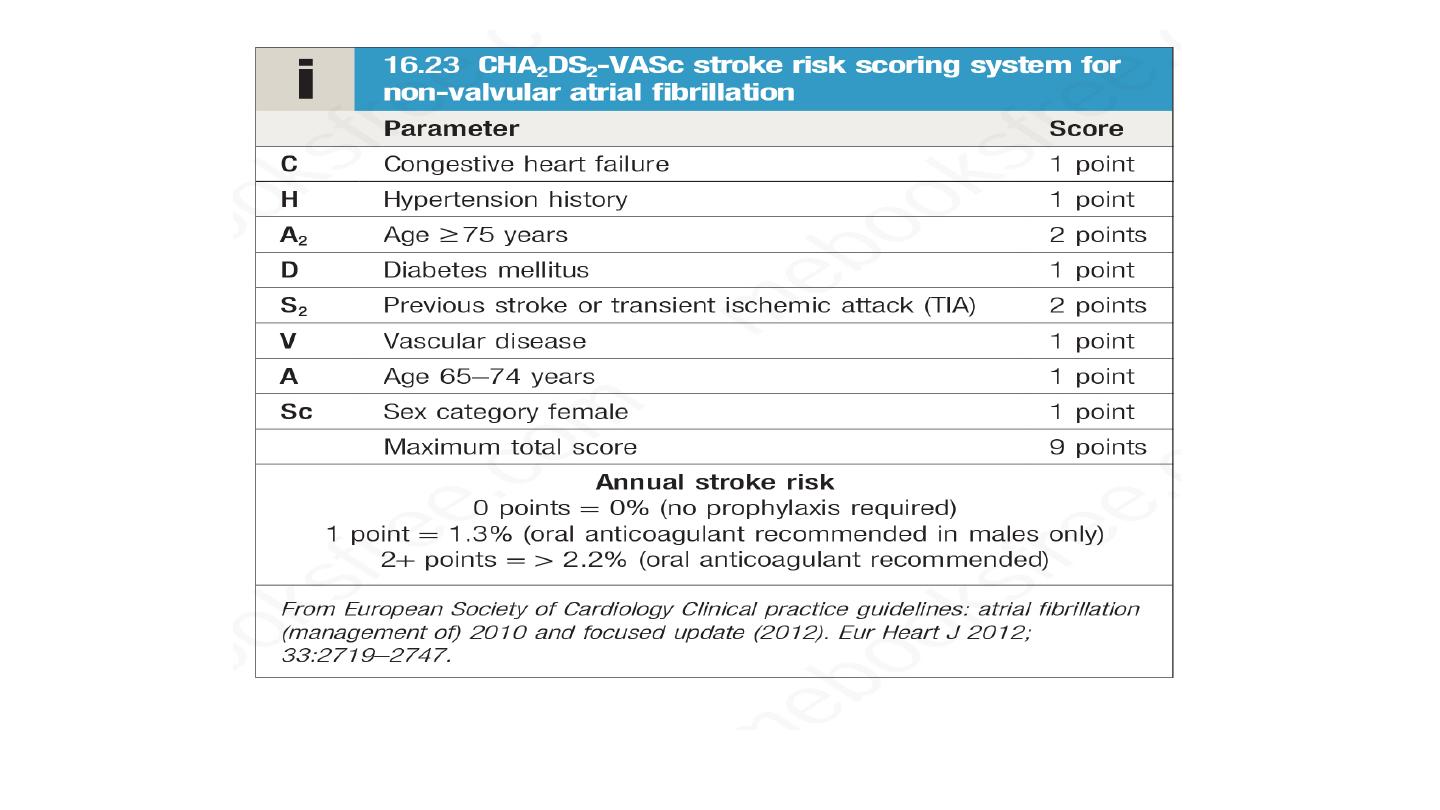
Valvular Af , anticoagulation with warfarin is indicated
Non valvular AF, CHADS VASC score is applied
patients with intermittent AF, stroke risk is similar to that in
patients with persistent AF when adjusted for CHA2DS2-VASc
score. The risk of embolism is only weakly related to the
frequency and duration of AF episodes, so stroke prevention
guidelines do not distinguish between those with paroxysmal,
persistent and permanent AF

The factor Xa inhibitors rivaroxaban, apixaban and edoxaban, and the
direct thrombin inhibitor dabigatran (collectively reffeted to as directly
acting oral anticoagulants, or DOACs), can be used as an alternative to
warfarin in non valvular AF.
They are at least as effective as warfarin at preventing thrombotic stroke
and are associated with a lower risk of intracranial haemorrhage. Other
advantages include the lack of requirement for monitoring and the fact
that they have fewer drug interactions.
Agents that reverse the effects of DOACs have been developed. These
include
idarucizumab
, which binds to dabigatran and allows acute bleeding
complications to be managed more effectively.
Aspirin should not be used since it has little or no effect on embolic stroke
and is associated with significant bleeding risk.

LA appendage occluder (watchman occluder)

Supraventricular tachycardia
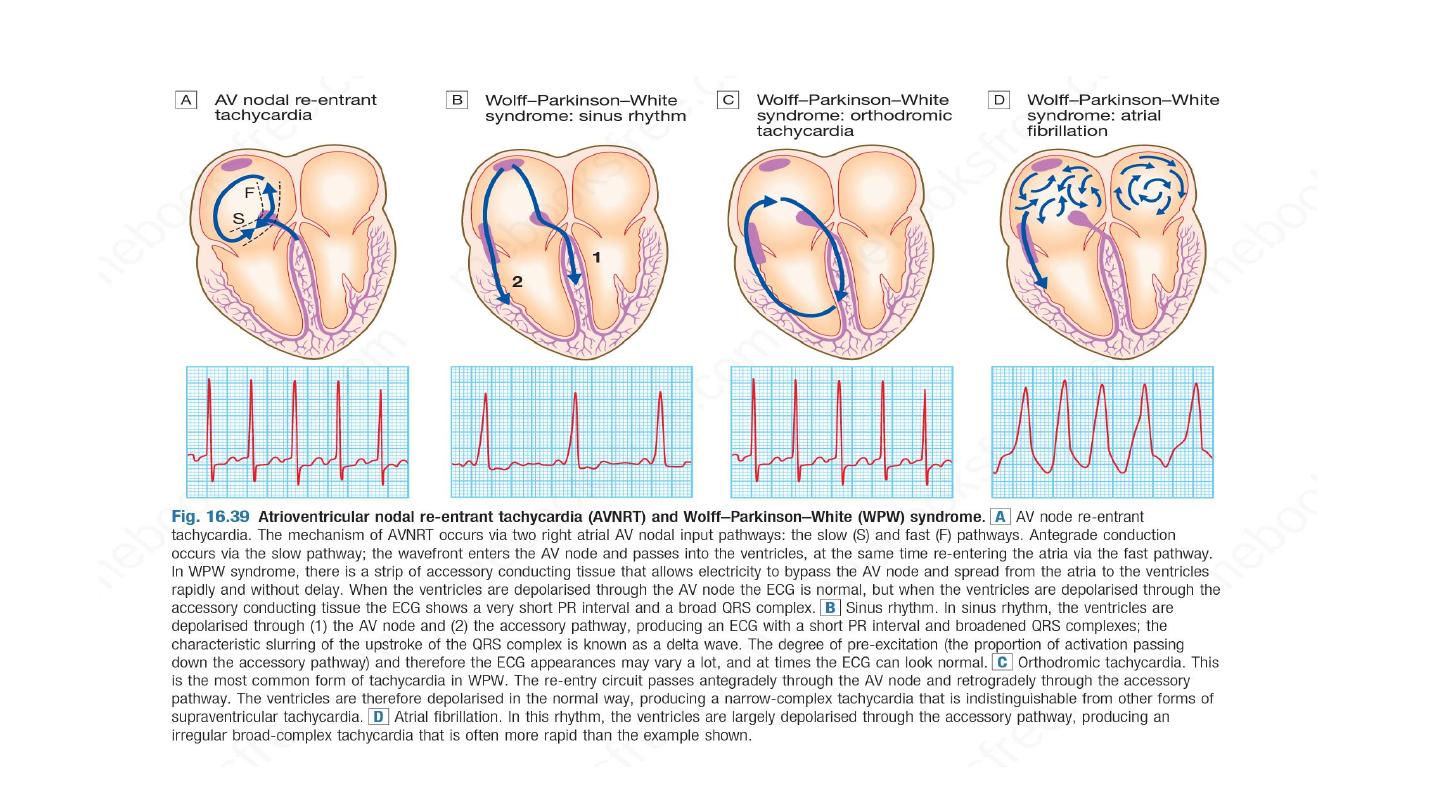
The three principal types are
atrioventricular nodal re-entrant tachycardia (AVNRT)
atrioventricular re-entrant tachycardia (AVRT)
atrial tachycardia.
The term SVT is technically incorrect as, in many cases, the ventricles
also form part of the re-entry circuit.

Atrioventricular nodal re-entrant tachycardia
AVNRT is a type of SVT caused by re-entry in a circuit involving the AV
node and its two right atrial input pathways: a superior ‘fast’ pathway and
an inferior ‘slow’ pathway.
Normal heart arrythmia, occur in Patients with structurally normal heart.
ECG : narrow complex, rate 120 to 240 bpm, rate dependent BBB may
occur
Present with palpitation lasting few seconds to hours

Management of AVNRT
For acute attack
Carotid sinus massage
Valsalva maneuver
Intravenous adenosine
Intrvenous verapamil
Intrvenous beta blocker , intravenous flecainide or amiodarone
For recurrent AVNRT, catheter ablation is indicated with success rate of
more than 90 %

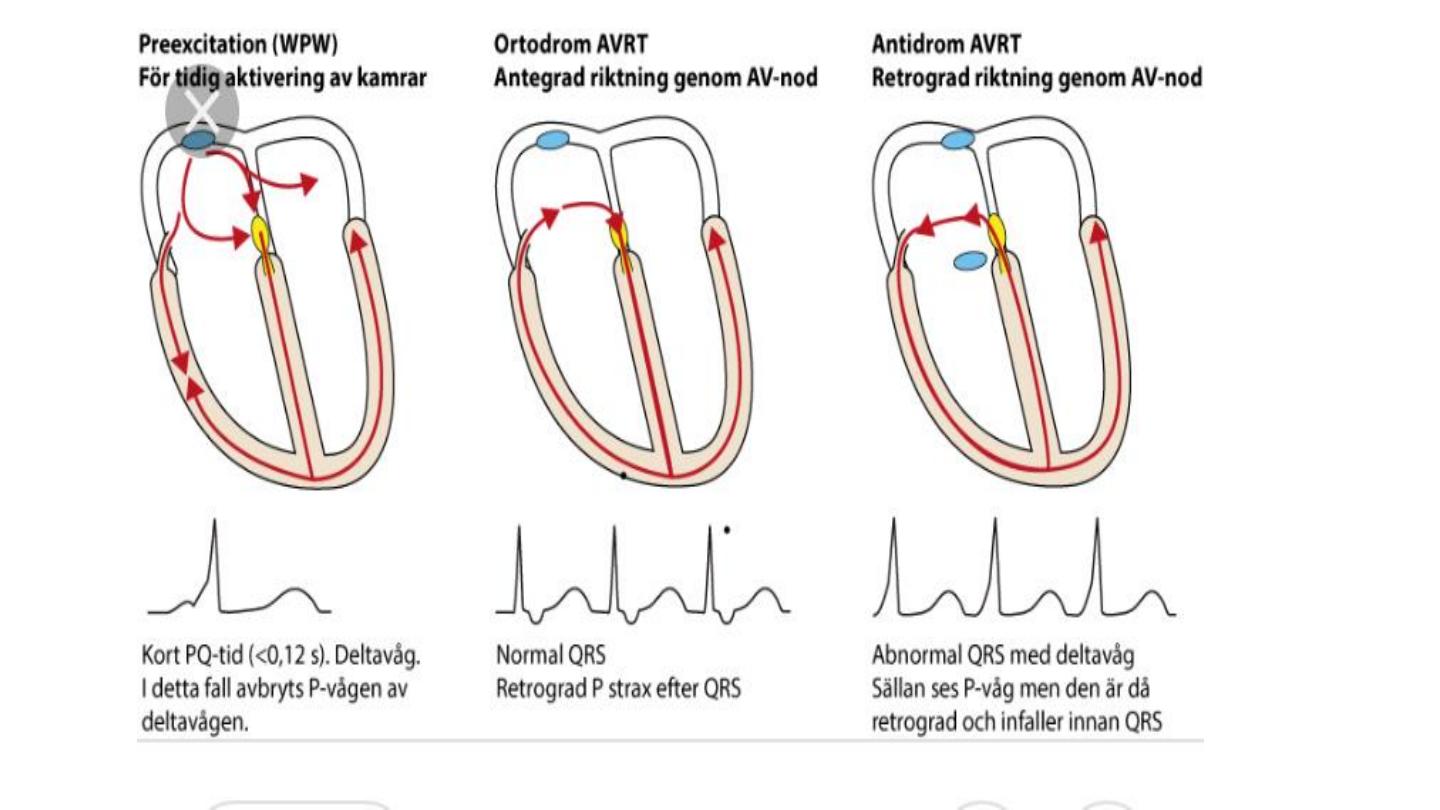
Atrioventricular re entry tavhycardia
In this condition there is an abnormal band of conducting tissue that connects
the atria and ventricles.
This so-called accessory pathway comprises rapidly conducting fibres that
resemble Purkinje tissue, in that they conduct very rapidly and are rich in sodium
channels.
In about 50% of cases, this pathway conducts only in the retrograde direction
(from ventricles to atria) and thus does not alter the appearance of the ECG in
sinus rhythm. This is known as a concealed accessory pathway.
In the rest, the pathway also conducts in an antegrade direction (from atria to
ventricles), so AV conduction in sinus rhythm is mediated via both the AV node
and the accessory pathway, distorting the QRS complex.

Premature ventricular activation through the accessory pathway
shorten the PR interval and result in initial slurring of QRS called delta
wave, this is called manifested pathway.
Wpw pattern, WPW syndrome ?
Accessory pathway and the AV node has different refractory period
and conduction velocity lead to re entry circuit

Managment
Narrow complex arrhythmia, retrograde conduction the accessiry pathway , i.v
adenosine and carotid sinus massage
Wide complex arrhythmia, antegrade conduction through AV node , class I c
antiarrhythmic with beta blocker
If AF occurs, it may produce a dangerously rapid ventricular rate because the
accessory pathway lacks the rate-limiting properties of the AV node. This is
known as pre-excited atrial fibrillation and may cause collapse, syncope and even
death. It should be treated as an emergency, usually with DC cardioversion.
Catheter ablation sucessful in majority of cases and it is curative treatment.

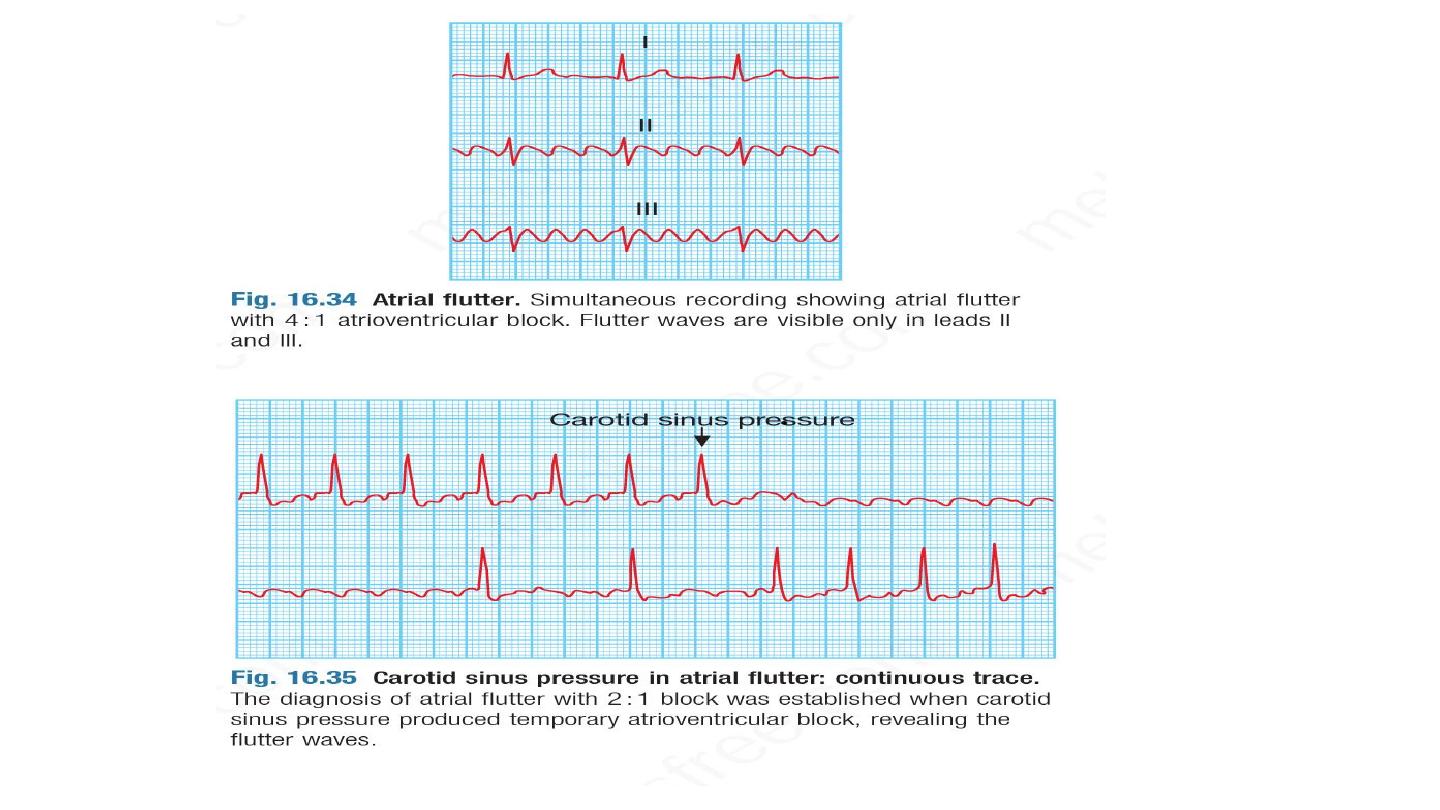
Atrial flutter
Atrial flutter is characterised by a large (macro) re-entry circuit,
usually within the right atrium encircling the tricuspid annulus.
The atrial rate is approximately 300/min, and is usually associated
with 2 : 1, 3 : 1 or 4 : 1 AV block (with corresponding heart rates of
150, 100 or 75/min).
Rarely, in young patients, every flutter wave is conducted, producing
a rate of 300/min and, potentially, haemodynamic compromise.
The ECG shows saw-tooth flutter waves. When there is regular 2 : 1 AV
block, it may be difficult to identify flutter waves that are buried in
QRS complexes and T waves.
Atrial flutter should always be suspected when there is a narrow-
complex tachycardia of 150/min. Carotid sinus pressure or intravenous
adenosine may help to establish the diagnosis by temporarily
increasing the degree of AV block and revealing flutter waves.

Managment
Medical management with digoxin, β-blockers or verapamil is often successful
in controlling the ventricular rate.
In many cases, however, it may be preferable to try to restore sinus rhythm by
direct current (DC) cardioversion.
Following cardioversion, β-blockers or amiodarone can be used to prevent
recurrent episodes of atrial flutter.
Class Ic anti-arrhythmic drugs such as flecainide are contraindicated because
there is a risk of slowing the flutter circuit, facilitating 1 : 1 AV nodal
conduction and producing a paradoxical extreme tachycardia and
haemodynamic compromise
.
Catheter ablation is a highly effective treatment, offering a greater than 90%
chance of complete cure, and is the treatment of choice for patients with
persistent symptoms. Anticoagulant management in patients with atrial flutter,
including management around cardioversion, is identical to that of patients with
atrial fibrillation.

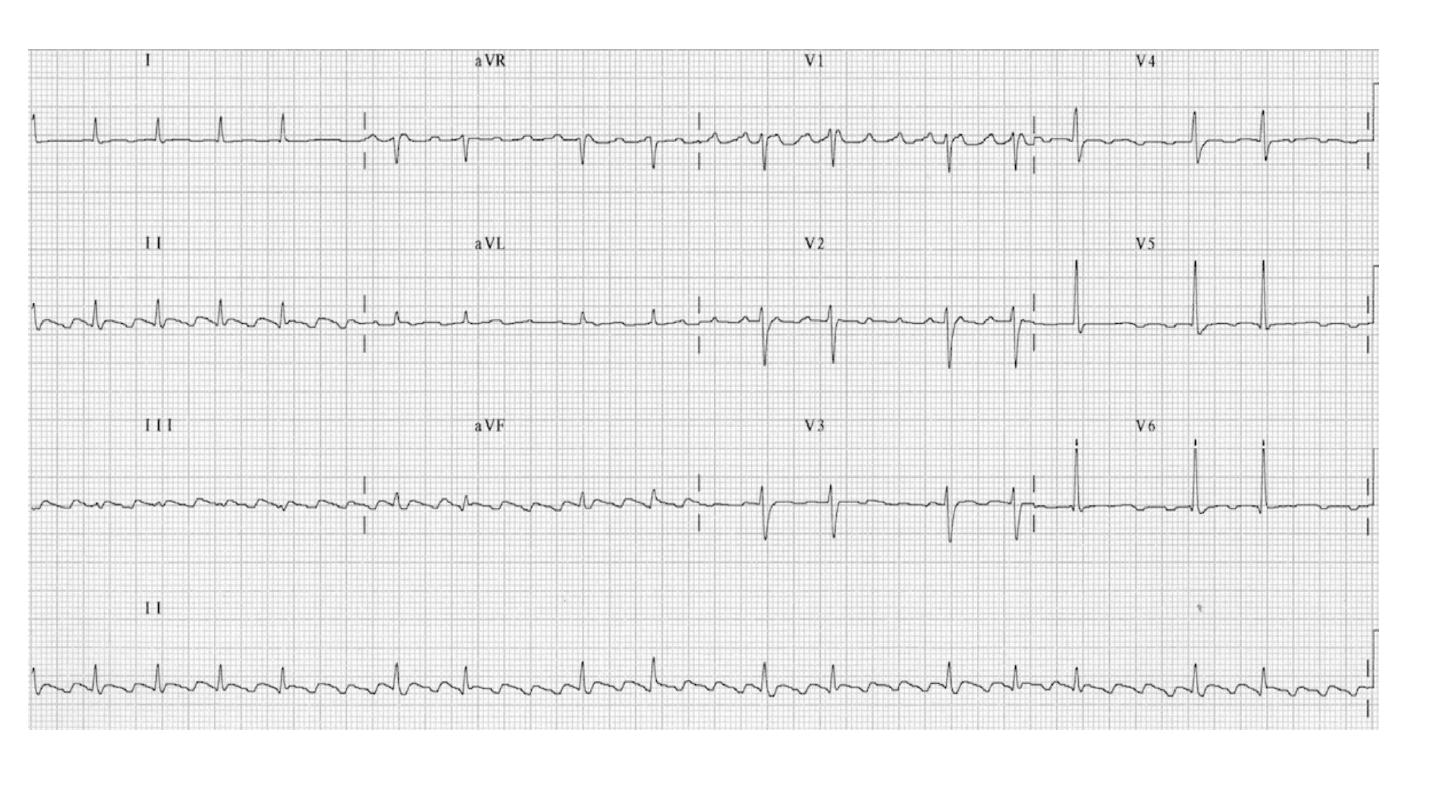
Premature ventricular ectopics
Ventricular premature beats (VPBs) are frequently found in healthy people
and their prevalence increases with age.
Ectopic beats in patients with otherwise normal hearts are more
prominent at rest and disappear with exercise.
Sometimes VPBs are a manifestation of subclinical coronary artery disease
or cardiomyopathy but also may occur in patients with established heart
disease following an MI. Most patients with VPBs are asymptomatic but
some present with an irregular heart beat, missed beats or abnormally
strong beats, due to increased cardiac output of the post-ectopic sinus
beat.
The ECG shows broad and bizarre complexes because the ventricles are
activated sequentially rather than simultaneously.
The complexes may be
unifocal
(identical beats arising from a single
ectopic focus) or multifocal (varying morphology with multiple foci, Fig.
16.40). ‘
Couplet
’ and ‘
triplet
’ are the terms used to describe two or three
successive ectopic beats. A run of alternating sinus and ventricular ectopics
called
bigeminy
.

Management of PVE
In symptomatic patients , b blocker helpful to control symptoms.
In refractory cases , catheter ablation can be useful.
Frequent PVCs are common in patients with structural heart disease,
like MI, cardiomyopathy, and associated with worse prognosis,
treatment should be directed toward underling heart disease,
unfortunately antiarrythmic medication not improve prognosis except
beta blocker in the setting of MI and heart failure.

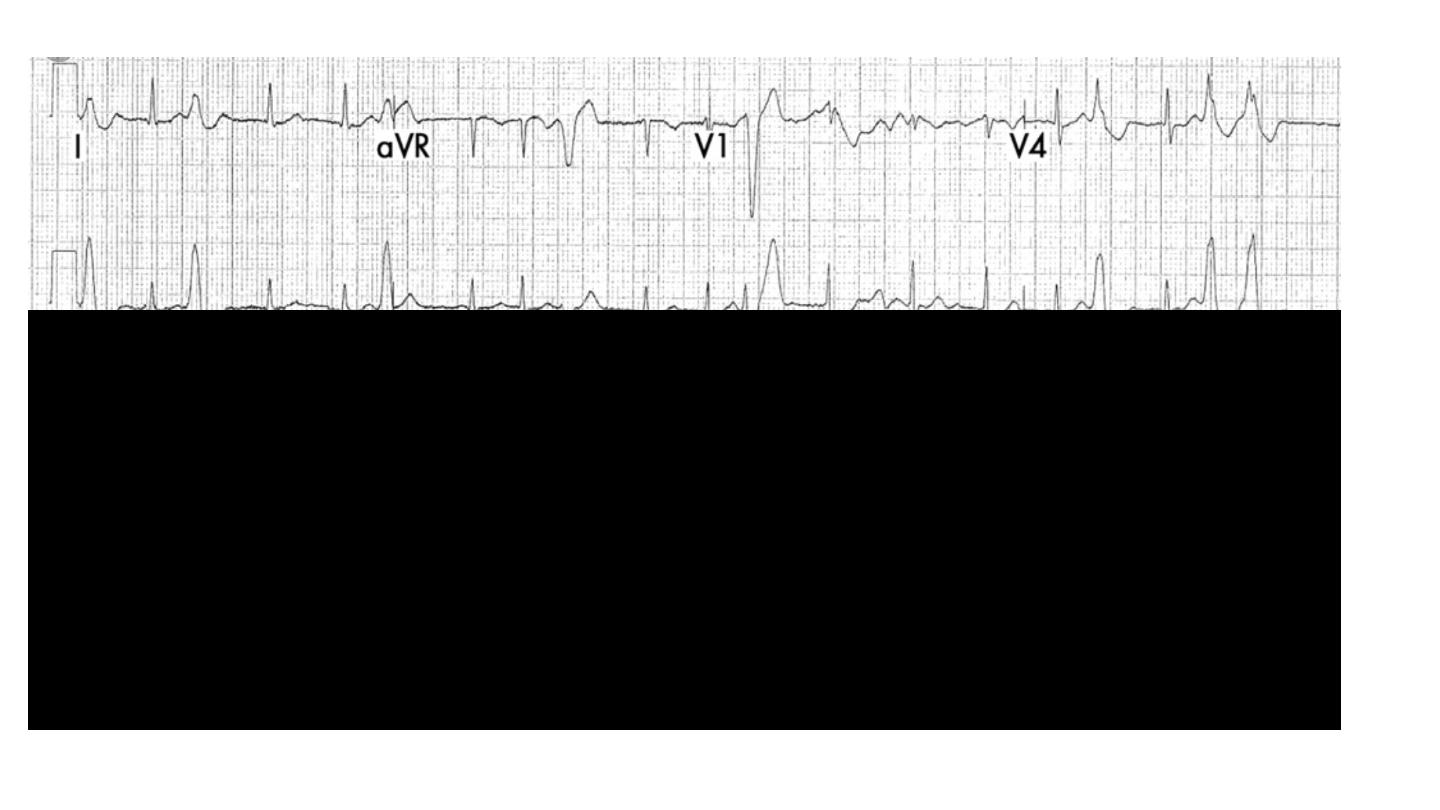
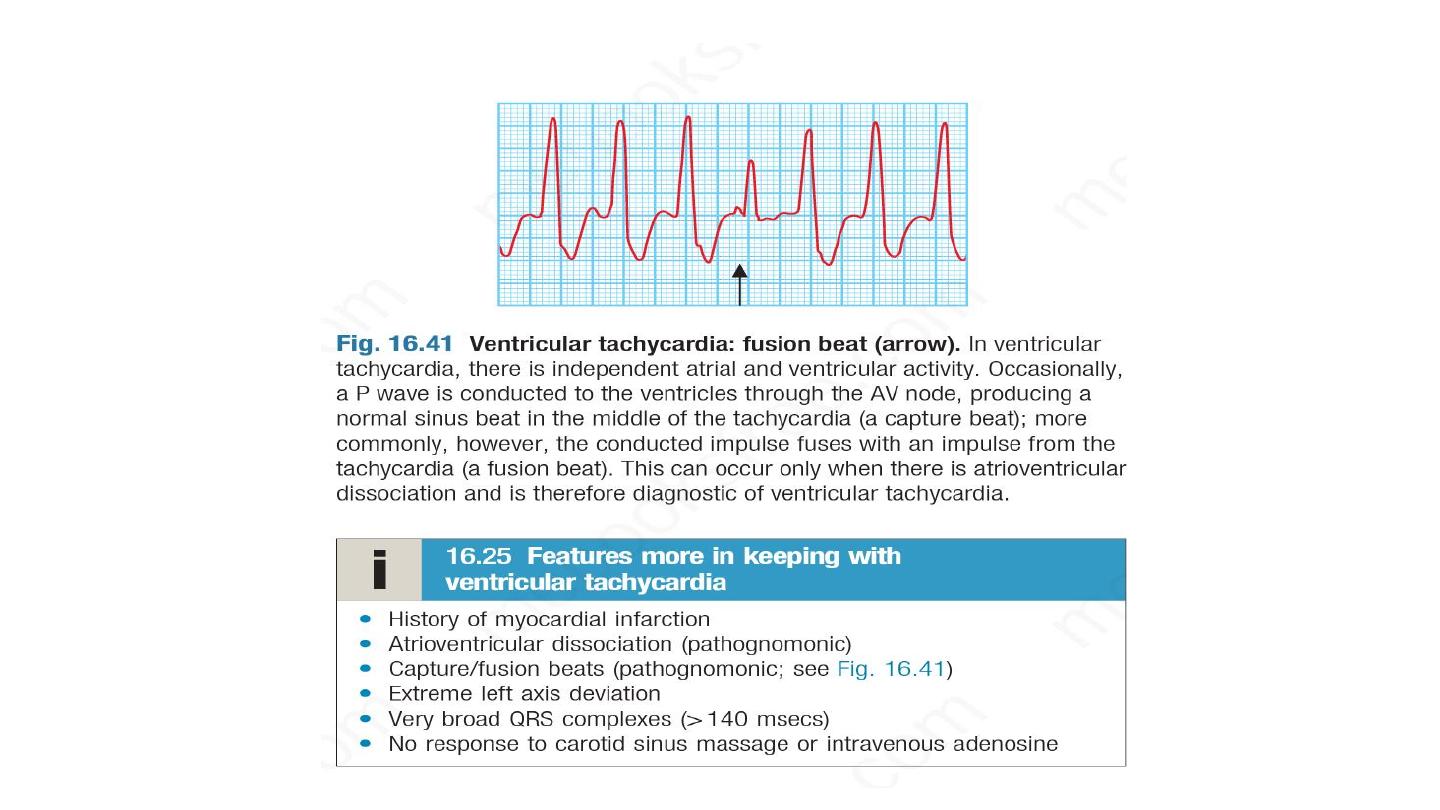
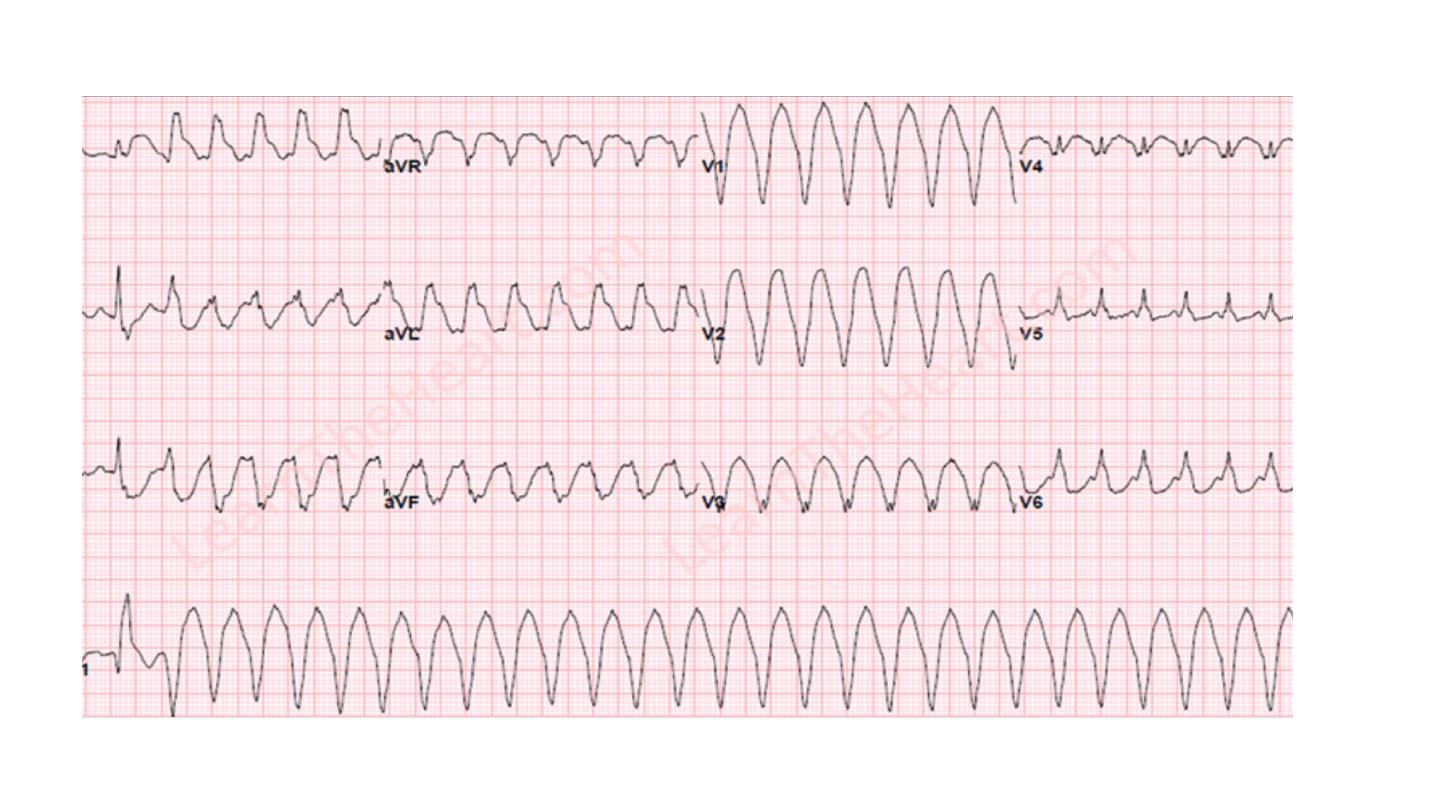
Ventricular tachycardia
vetricular tachycardia (VT) occurs most commonly in the settings of acute MI,
chronic coronary artery disease and cardiomyopathy , It is associated with
extensive ventricular disease, impaired left ventricular function and ventricular
aneurysm.
In these settings, VT may cause haemodynamic compromise or degenerate into
ventricular fibrillation.
VT is caused by abnormal automaticity or triggered activity in ischaemic tissue,
or by re-entry within scarred ventricular tissue. Patients may complain of
palpitation or symptoms of low cardiac output, including dyspnoea,
lightheadedness and syncope.
The ECG shows tachycardia and broad, abnormal QRS complexes with a rate of
more than 120/min.
It may be difficult to distinguish VT from SVT with bundle branch block or pre-
excitation (WPW syndrome) on ECG but features in favour of VT are listed in Box
16.25. A 12-lead ECG or electrophysiology study (p. 454) may help establish the
diagnosis.

Patients recovering from MI sometimes have periods of
idioventricular rhythm (‘slow’ VT) at a rate only slightly above the
preceding sinus rate and below 120/min.
These episodes often reflect reperfusion of the infarct territory and
may be a good sign. They are usually self-limiting and asymptomatic,
and do not require treatment.
Occasionally, VT occurs in patients with otherwise healthy hearts
(‘normal heart VT’), usually because of abnormal automaticity in the
right ventricular outflow tract or one of the fascicles of the left bundle
branch
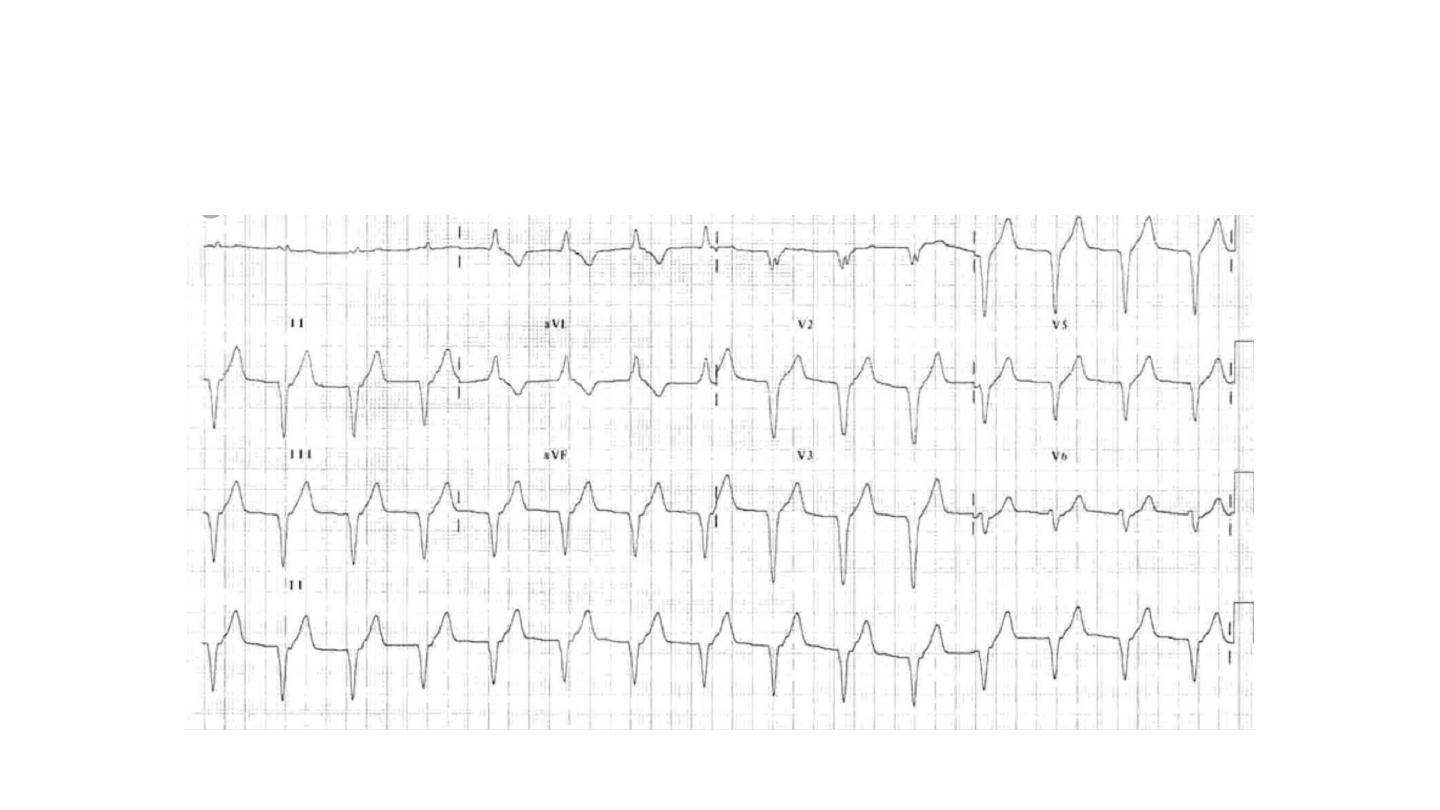
Idioventricular rhythm

Managment
If VT associated with hemodynamic instability, require DC cardioversion (
chest pain, dizziness, raised JVP, bibasal crackles)
If VT is hemodynamically stable, intravenous amiodarone bolus and
maintenance . Intravenous lidocaine can be used for chemical
cardioversion of VT , but might cause hypotension, convulsion, myocardial
depression.
VT can be prevented by beta blocker and amiodarone, class IC
antiarrythmic is contraindicated in patient with structural heart disease like
CAD, cardiomyopathy.
Consider ICD implantation

Torsade de pointes
This form of polymorphic VT is a complication of prolonged
ventricular repolarisation (prolonged QT interval).
The ECG shows rapid irregular complexes that seem to twist
around the baseline as the mean QRS axis changes.
The arrhythmia is usually non-sustained and repetitive, but may
degenerate into ventricular fibrillation.
During periods of sinus rhythm, the ECG will usually show a
prolonged QT interval (> 0.44 sec in men, > 0.46 sec in women
when corrected to a heart rate of 60 bear per minutes)
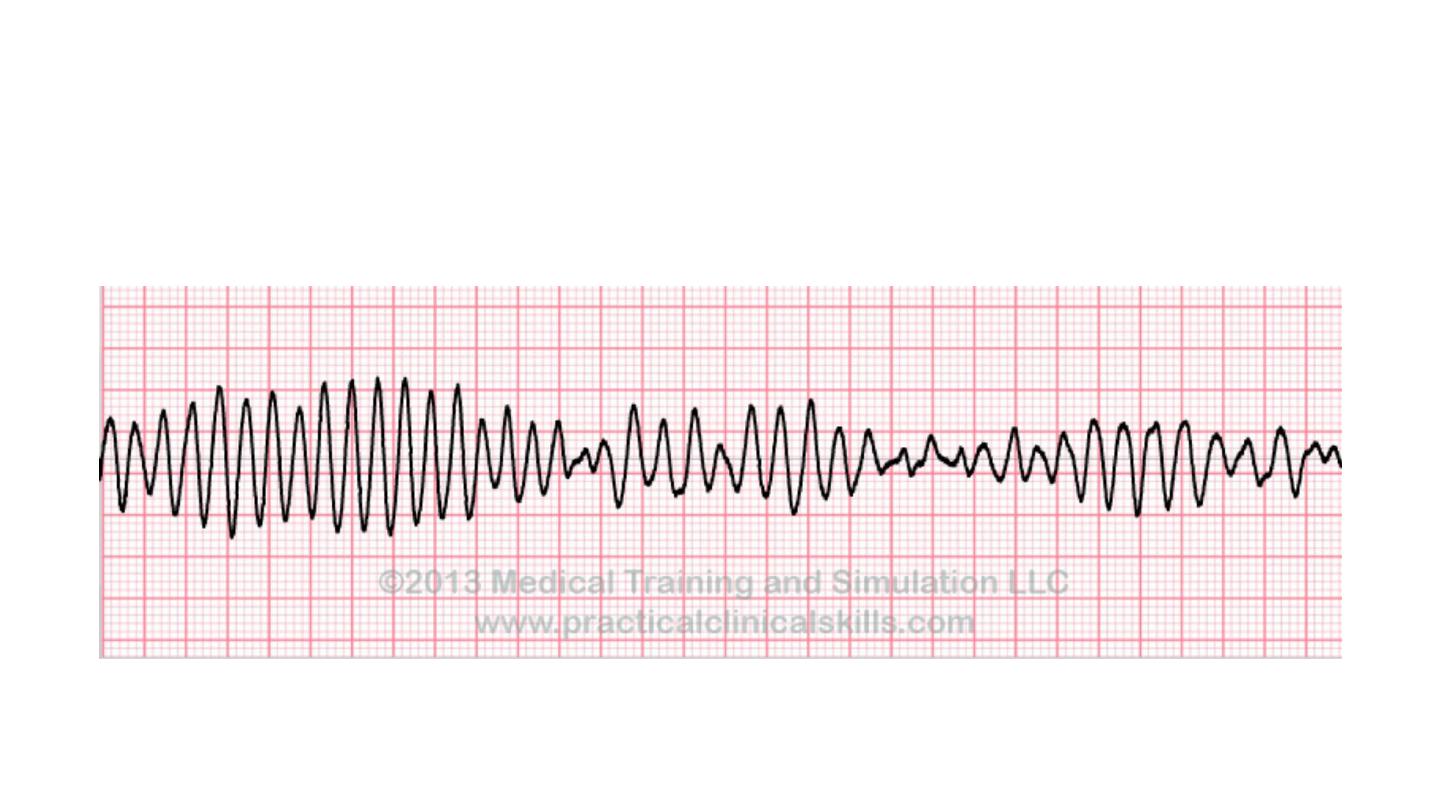
Torsade de pointes

The arrhythmia is more common in women and is often
triggered by a combination of factors, such as administration of
QT-prolonging medications and hypokalaemia.
The congenital long QT syndromes are a family of genetic
disorders that are characterised by mutations in genes that code
for cardiac sodium or potassium channels.
Adrenergic stimulation through vigorous exercise is a common
trigger in long QT type 1, and a sudden noise may trigger
arrhythmias in long QT type 2. Arrhythmias are more common
during sleep in type 3


Managment
Intravenous magnesium 8 mmol over 15 mins, 72 mmol over 24
hours should be given in all patients with torsade de pointes.
Overdrive atrial pacing useful suppress arrhythmia through rate
dependent shortening of QT interval.
Intravenous isoprenaline is alternative to atrial pacing , but
contraindicated in long QT syndrome.

Beta-blockers are effective at preventing syncope in patients with
congenital long QT syndrome.
Some patients, particularly those with extreme QT interval
prolongation (> 500 msecs) or certain high-risk genotypes, should
be considered for an implantable defibrillator.
Left stellate ganglion block may be of value in patients with
resistant arrhythmias.

Brugada syndrome
Brugada syndrome is a related genetic disorder that may present
with polymorphic VT or sudden death.
It is characterised by a defect in sodium channel function and
an abnormal ECG (right bundle branch block and ST elevation in
V1 and V2 but not usually prolongation of the QT interval).
The only known effective treatment is an implantable
defibrillaton
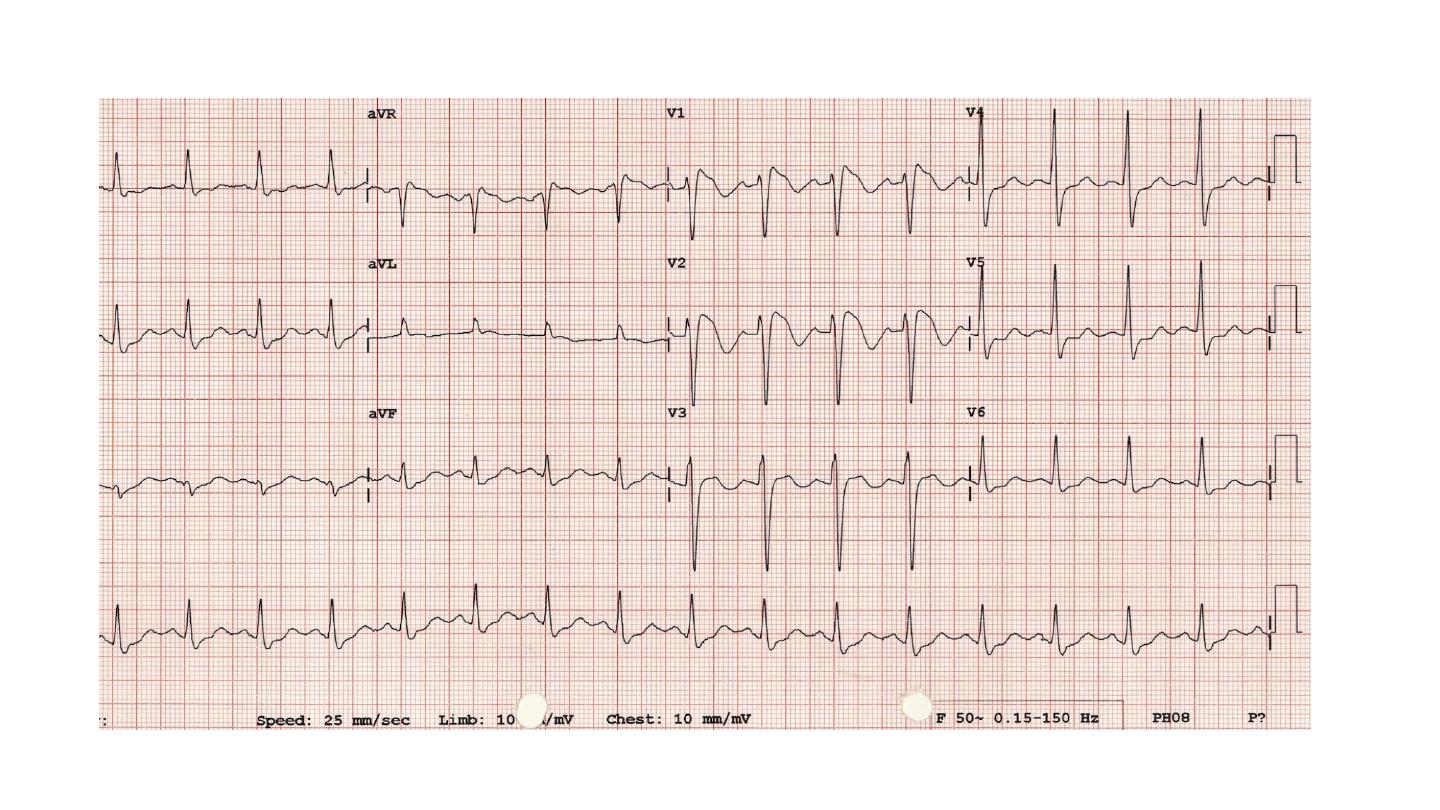

Atrioventricular block
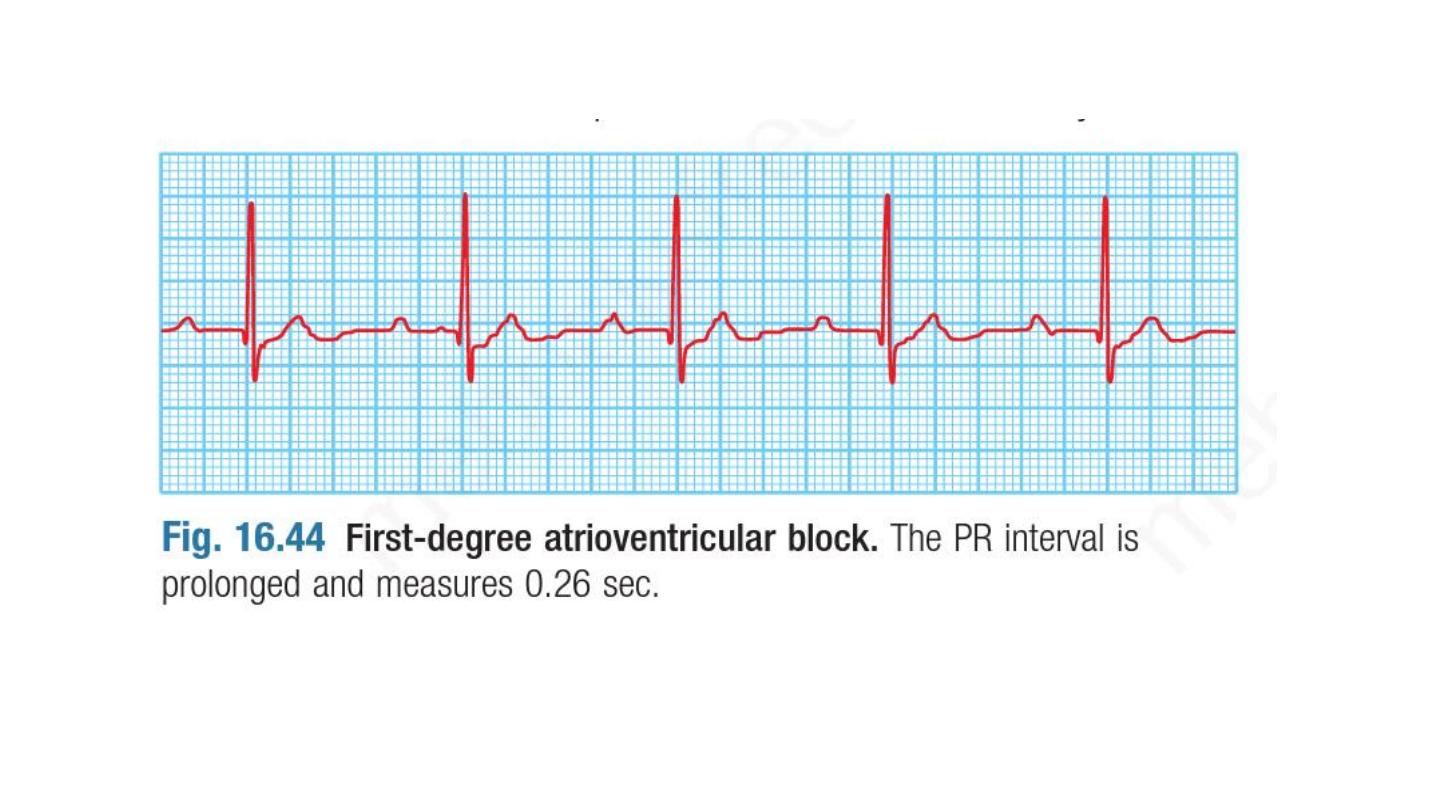
First-degree atrioventricular block .In this condition, AV
conduction is delayed and so the PR interval is prolonged (> 0.20
sec. It rarely causes symptoms and does not usually require
treatment.
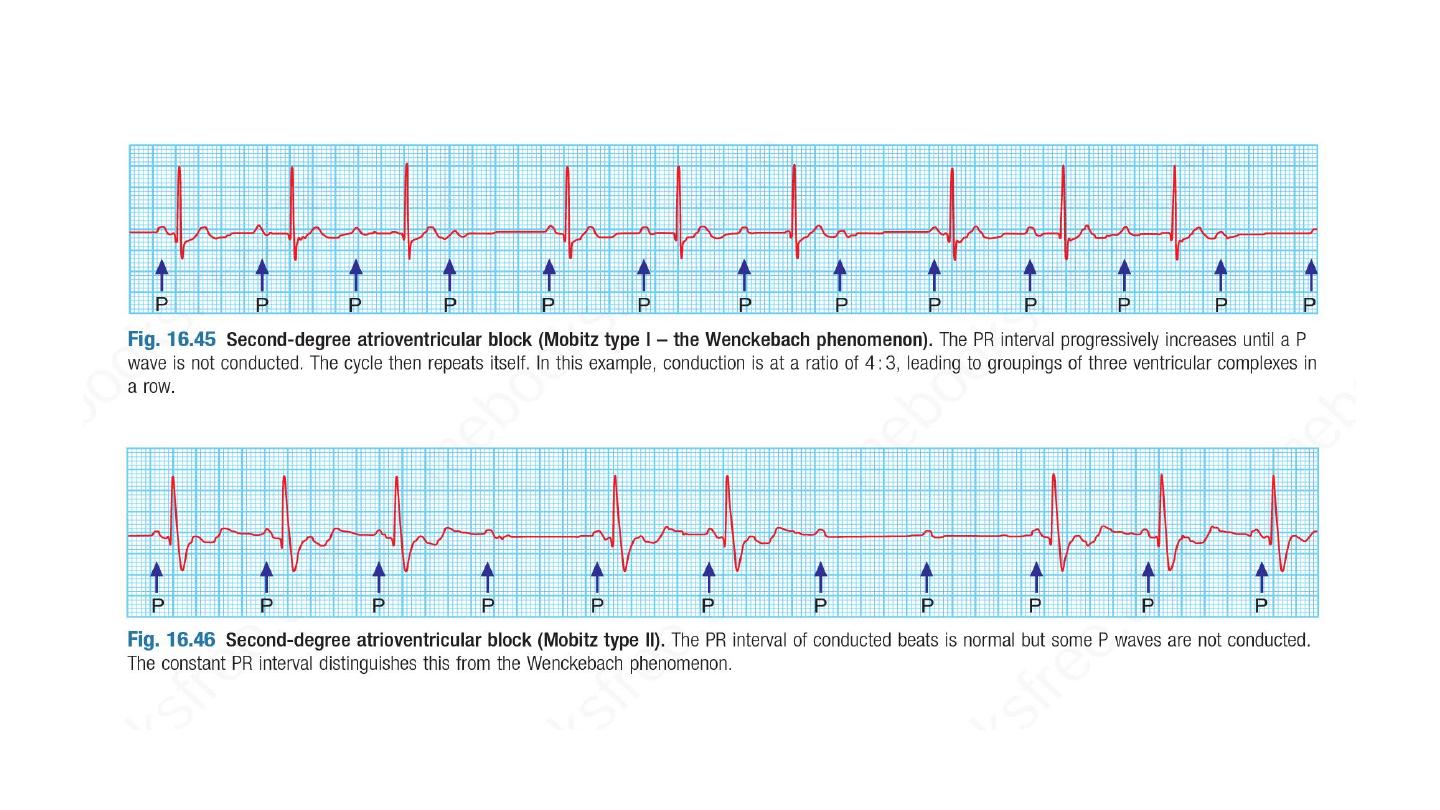
Second-degree atrioventricular block Here dropped beats occur
because some impulses from the atria fail to conduct to the
ventricles. Two subtypes are recognized :

In Mobitz type I second-degree AV block, there is progressive
lengthening of successive PR intervals, culminating in a dropped
beat.
The cycle then repeats itself. This is known as the Wenckebach
phenomenon and is usually due to impaired conduction in the
AV node itself. The phenomenon may be physiological and is
sometimes observed at rest or during sleep in athletic young
adults with high vagal tone.
In Mobitz type II second-degree AV block, the PR interval of the
conducted impulses remains constant but some P waves are not
conducted. This is usually caused by disease of the His–Purkinje
system and carries a risk of asystole.

Third degree atrioventricular block :
In third-degree AV block, conduction fails completely and the atria and
ventricles beat independently. This is known as AV dissociation, Ventricular
activity is maintained by an escape rhythm arising in the AV node or bundle
of His (narrow QRS complexes) or the distal Purkinje tissues (broad QRS
complexes).
Distal escape rhythms tend to be slower and less reliable. Complete AV
block (Box 16.27) produces a slow (25–50/min), regular pulse that does not
vary with exercise, except in the case of congenital complete AV block.
There is usually a compensatory increase in stroke volume, producing a
large-volume pulse. Cannon waves may be visible in the neck and the
intensity of the first heart sound varies due to the loss of AV synchrony.
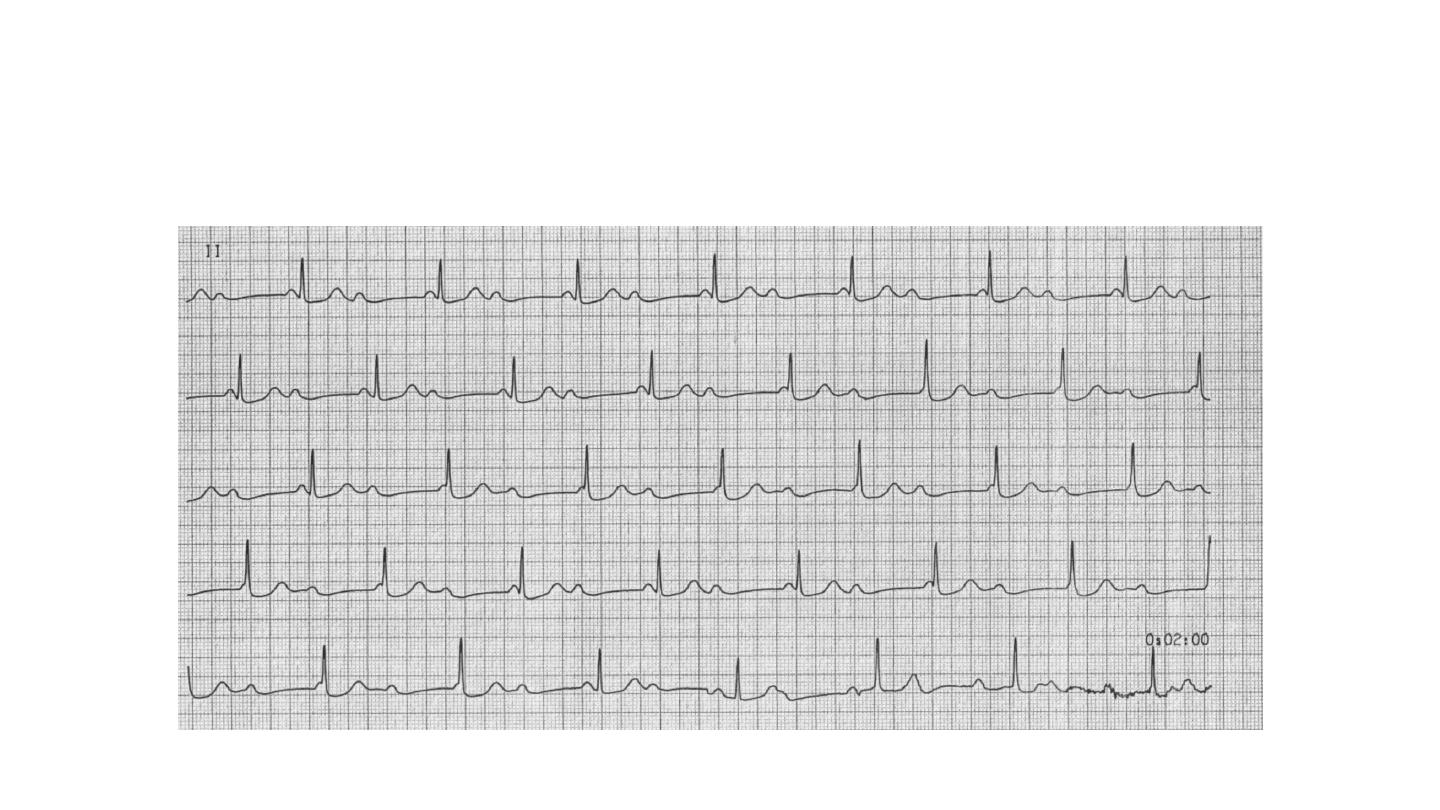
CHB


Clinical features
• Syncope
• Adam stoke attacks is a sudden loss of consciousness that occur
without warning and result in collapse. Brief anoxic seizure may
developed if there is a prolong asystole. There is pallor and death like
appearance during the attack , and when the heart beat again there is
characteristic flushing.

Treatment of heart block
1
st
and mobitz type 1 second degree AV block, usually asymptomatic
Mobitz type 2 second degree AV block , 3
rd
degree AV block; Acute
inferior MI is often complicated by transient AV block because
the right coronary artery (RCA) supplies the AV node. There is
usually a reliable escape rhythm and, if the patient remains well,
no treatment is required. Symptomatic second- or third-degree
AV block may respond to atropine (0.6 mg IV, repeated as
necessary) or, if this fails, a temporary pacemaker. In most cases,
the AV block will resolve within 7–10 days

Second- or third-degree AV heart block complicating acute
anterior MI indicates extensive ventricular damage involving both
bundle branches and carries a poor prognosis.
Mobitz type 2 or third degree AV block , even a symptomatic , if not
due to reversible cause is an indication for pacing.

Lines in the management of arrythmias
Drugs
DC shock ( cardioversion and defibrillation)
Implantable device like pacemaker and ICD
Catheter ablation
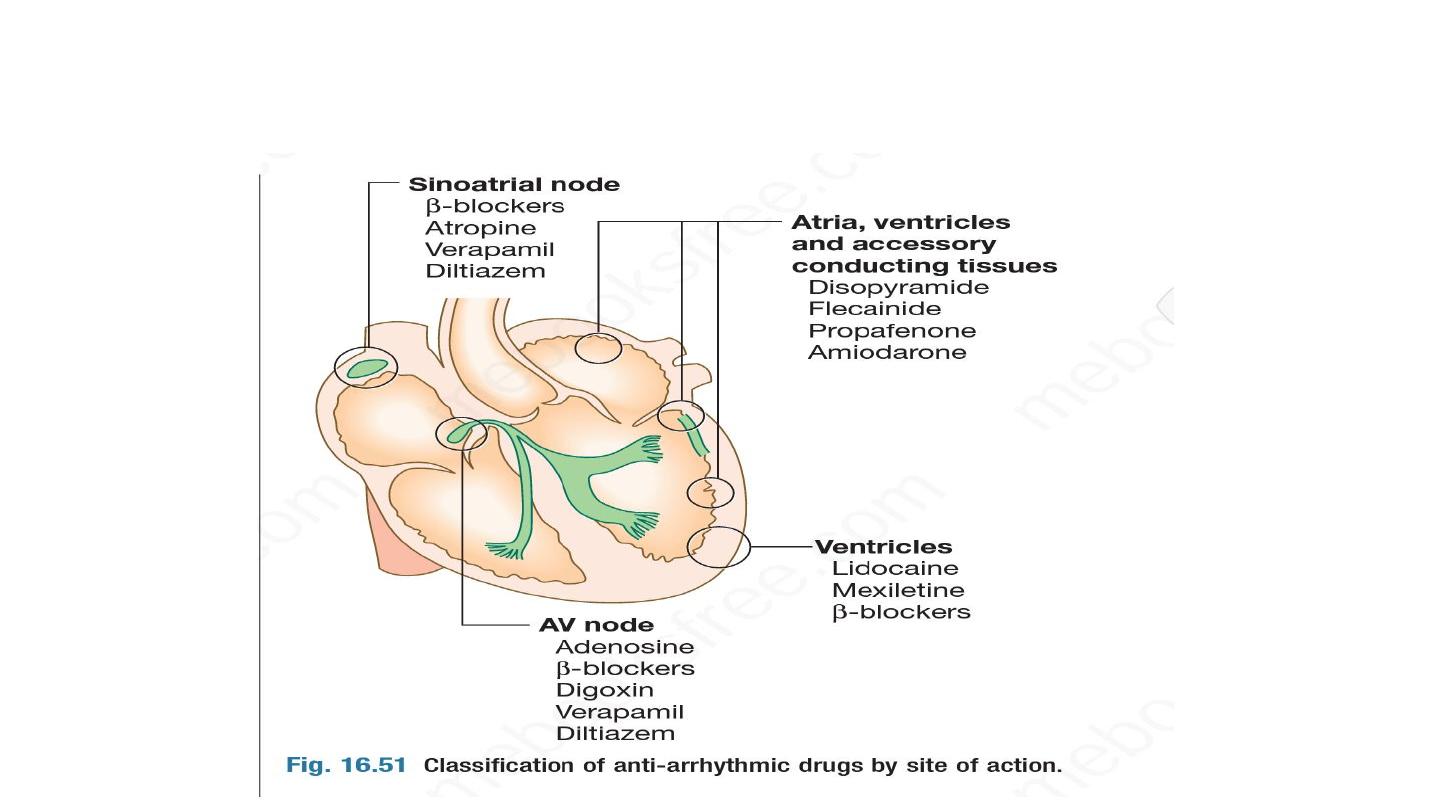
Antiarrythmic medication

