
Malignant tumours of the ovary
Dr.Nadia Mudher Al-Hilli
FICOG
Department of Obs&Gyn
College of Medicine
University of babylon

Objectives of lecture
• Learn how malignant disease of the ovary
presents.
• Learn how ovarian cancer is investigated and
staged.
• Learn how ovarian cancer is managed.

Epidemiology:
• The lifetime risk is 1.4 %
• Mean age of presentation is 64 years.
• More prevalent in developed nations
• incidence vary with ethnicity, Caucasian have higher
incidence than Asian.
• large proportion of ovarian cancers may originate in
the Fallopian tube, rather than the ovarian surface
epithelium.
• There is a significant genetic aspect to ovarian cancer.
• hereditary cancer present early, with a mean age at
diagnosis of 54 years.
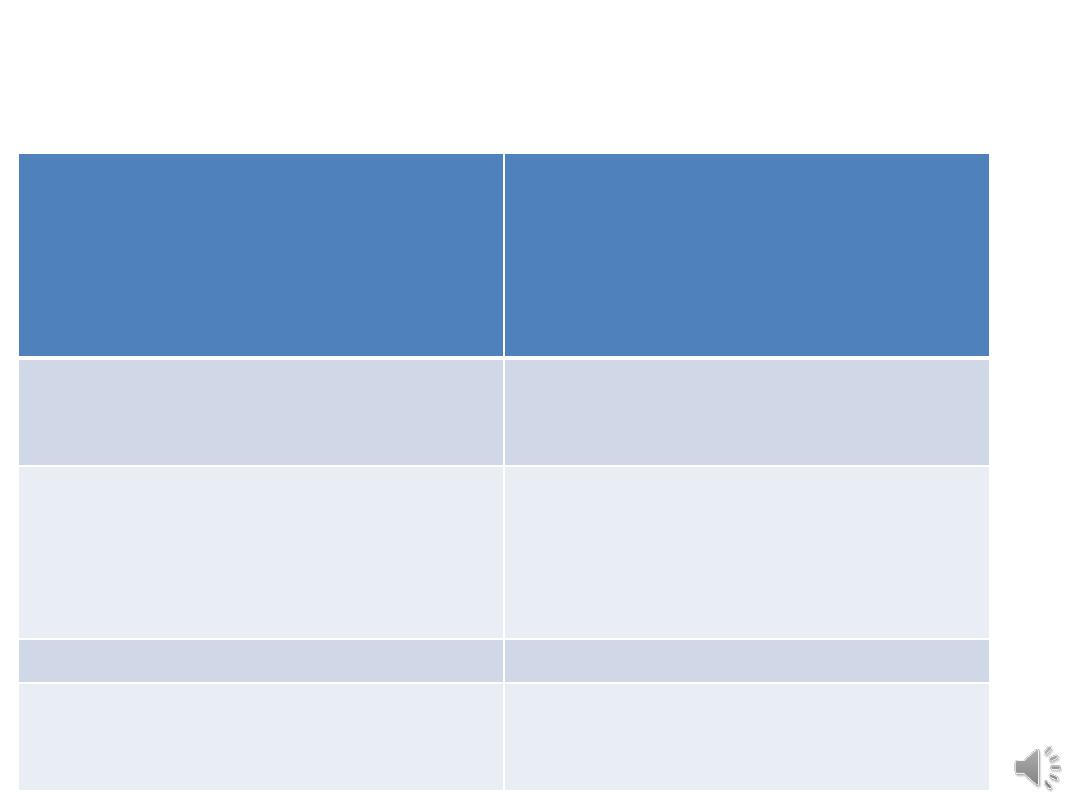
Classification of malignant ovarian tumours
1 Epithelial ovarian tumours (80%)
High-grade serous (75%)
Endometrioid
Clear cell
Mucinous
Low-grade serous
(Borderline)
2 Sex cord stromal tumours (10%)
Granulosa cell
Sertoli–Leydig
Gynandroblastoma
3 Germ cell tumours (10%)
Dysgerminoma
Endodermal sinus (yolk sac)
Teratoma
Choriocarcinoma
Mixed
4 Metastatic (including Krukenberg
tumours
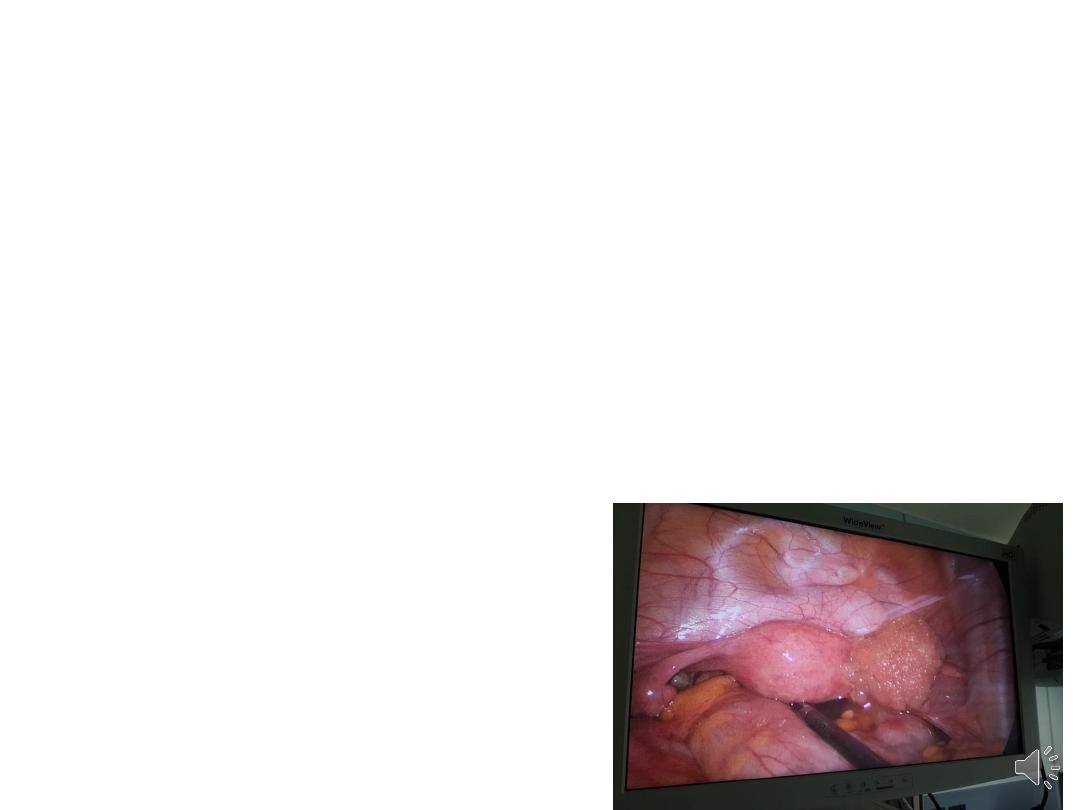
Borderline epithelial tumours:
• well differentiated, with some features of
malignancy (nuclear pleomorphism, cellular
atypia) but do not invade the basement
membrane.
• constitute 10% of epithelial ovarian tumours.
• May spread to pelviabdominal structures but not
recur after initial
surgery.
• The majority of BOTs are
serous tumours.
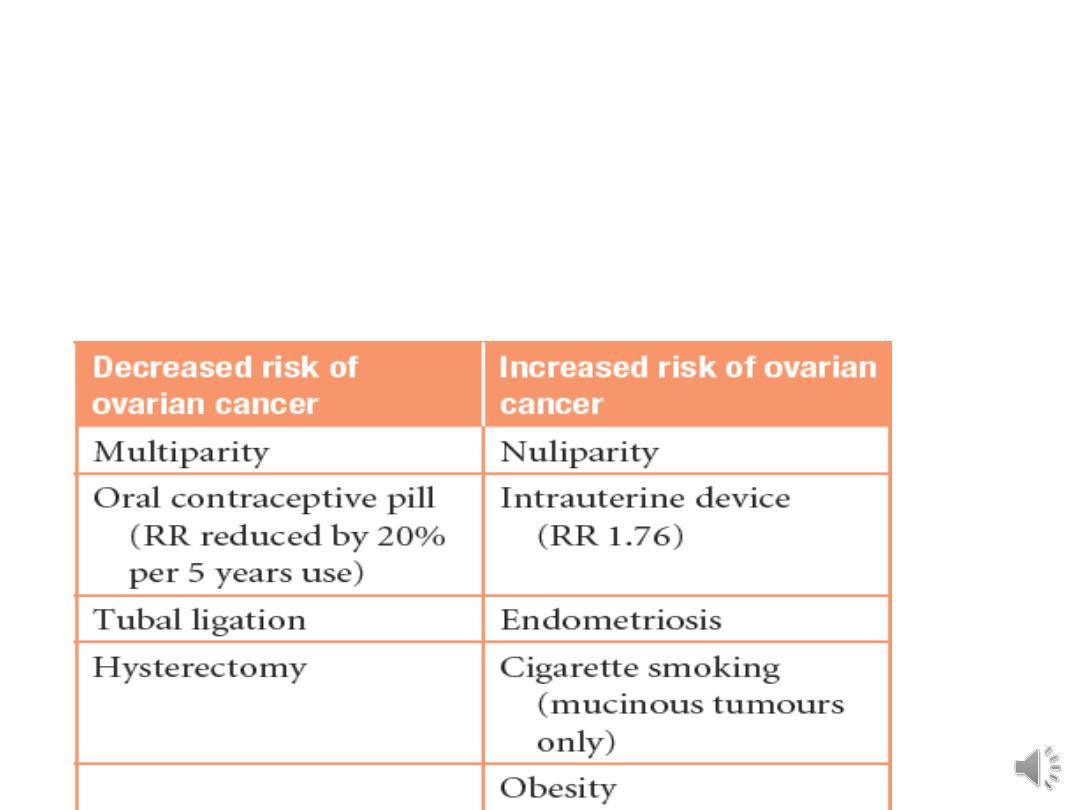
Etiology of Epithelial tumours:
• Incessant ovulation theory:
• Subfertility treatment:
• Genetic factors:
Risk factors in ovarian cancer:

High-grade pelvic serous carcinomas:
• most present with advanced disease involving the ovary,
Fallopian tube and peritoneal surfaces, making it
impossible to establish the anatomical site of origin.
• This term incorporate all high-grade serous tumours arising
from the ovary, Fallopian tube and/or peritoneum.
• New data suggest a Fallopian tubal precursor lesion for
high-grade pelvic serous tumours. These precursors are
called serous tubal intraepithelial carcinoma (STIC) lesions.

Familial ovarian cancer:
• Constitute 10–15 % of epithelial ovarian cancer.
• mutations in BRCA1, BRCA2 and Lynch syndrome
• The lifetime risk rises to 5 % if one family member
affected and increases to 40–50 % if two first
degree relatives are affected.
• it occur 10 years earlier than sporadic cancers

• 90% of hereditary cancer is the breast ovarian
cancer syndrome (BRCA)
• The defective gene is most commonly the tumour-
suppressor gene BRCA1 (80%), BRCA2 (15%).
• Lynch syndrome is hereditary non-polyposis
colorectal cancer (HNPCC) and is associated with
endometrial cancer and a 10 per cent risk of
ovarian cancers.

Preventing ovarian cancer
• Women test positive for a BRCA mutation are offered risk-reducing
prophylactic BSO after completing their families.
• reduces risk of ovarian cancer by 90% and premenopausal breast
cancer by 50%.
• should be carried out prior to the age-related surge in ovarian cancer.
• bilateral salpingectomy with delayed oophorectomy in the 30s and
early 40s may offset the morbidity associated with a surgical
menopause in young women.
• opportunistic removal of the Fallopian tubes during hysterectomy for
benign indications, tubal ligation (sterilization) and hysterectomy
with ovarian conservation, also reduces ovarian cancer risk.
• Chemoprevention using COCP reduces ovarian cancer risk by up to
50%.

Screening:
• Screening using transvaginal ultrasound scan
(TVUSS) and CA125 measurement has not
been shown to improve survival in women
with a familial predisposition to ovarian
cancer.
• because the high grade serous tumours that
are associated with BRCA mutation carrier
status develop rapidly and most are at an
advanced stage before they can be picked up
by screening.

Clinical features of epithelial ovarian
cancer
• symptoms are nonspecific and often vague.
The difficulty with clinical diagnosis is the
main reason that patients with ovarian
carcinoma present with late stage disease

Common symptoms are:
• persistent pelvic and abdominal pain
• increased abdominal size/persistant bloating
• difficulty eating and feeling full quickly.
Other frequent symptoms:
• change in bowel habit
• urinary symptoms
• back ache
• irregular bleeding
• fatigue

Clinical Examination:
• Pelvic and abdominal examination may reveal a fixed, hard
mass arising from the pelvis. with or without ascites
• Chest examination is important to assess pleural fluid and
the neck and groin should be examined for enlarged nodes.
The differential diagnosis:
• Non epithelial ovarian cancer
• Tuboovarian abscess
• endometriomas
• fibroids

• TVUSS is the initial imaging modality of choice to check
for pelvic pathology.
• US characteristics:
• Size
• Consistency
• the presence of solid elements
• bilaterality
• ascites
• extraovarian disease, including peritoneal thickening
and omental deposits.
In conjunction with Ca125 measurement and menopausal
status, a risk of malignancy (RMI) is calculated.
Investigations:
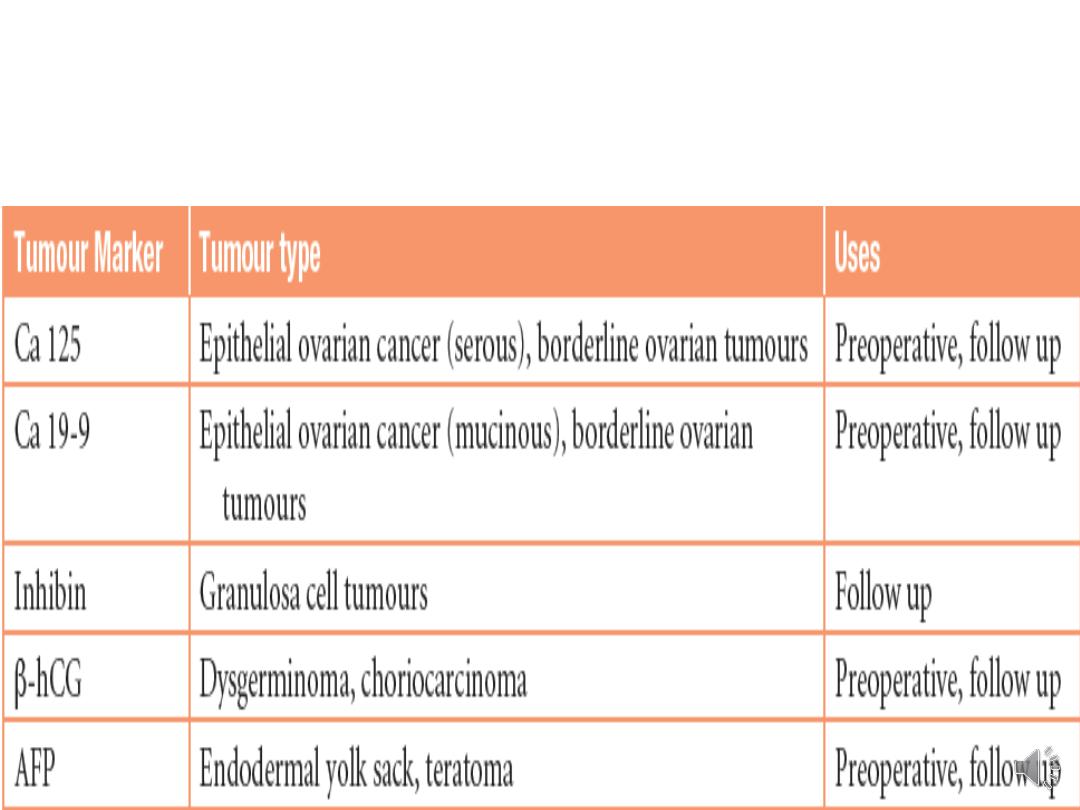
Tumour markers used in ovarian
carcinoma

• Pelvic pathology at intermediate or high risk of
malignancy is further imaged using computed
tomography (CT) for extrapelvic disease & staging
and/or magnetic resonance imaging (MRI) scans help
define tissue planes & operability.
Other investigations required for preoperative work-up
include:
• chest X-ray,
• electrocardiography (ECG)
• full blood count
• urea and electrolytes
• liver function tests.

Other investigations:
• Endometrial biopsy, especially if conservative surgery
is to be undertaken.
• If the patient presents with gross ascites or pleural
effusion, paracentesis or pleural aspiration may be
required for relief of symptoms or for diagnosis.
• barium enema or colonoscopy if bowel symptoms
are present or there is a possibility of a primary
colorectal tumour.

• Metastatic spread:
• pelvic peritoneum & pelvic organs are
involved by direct spread. malignant cells on
all intra-abdominal structure surfaces.
• Lymphatic spread involve pelvic & para-aortic
lymph nodes, nodes in the neck & inguinal
region.
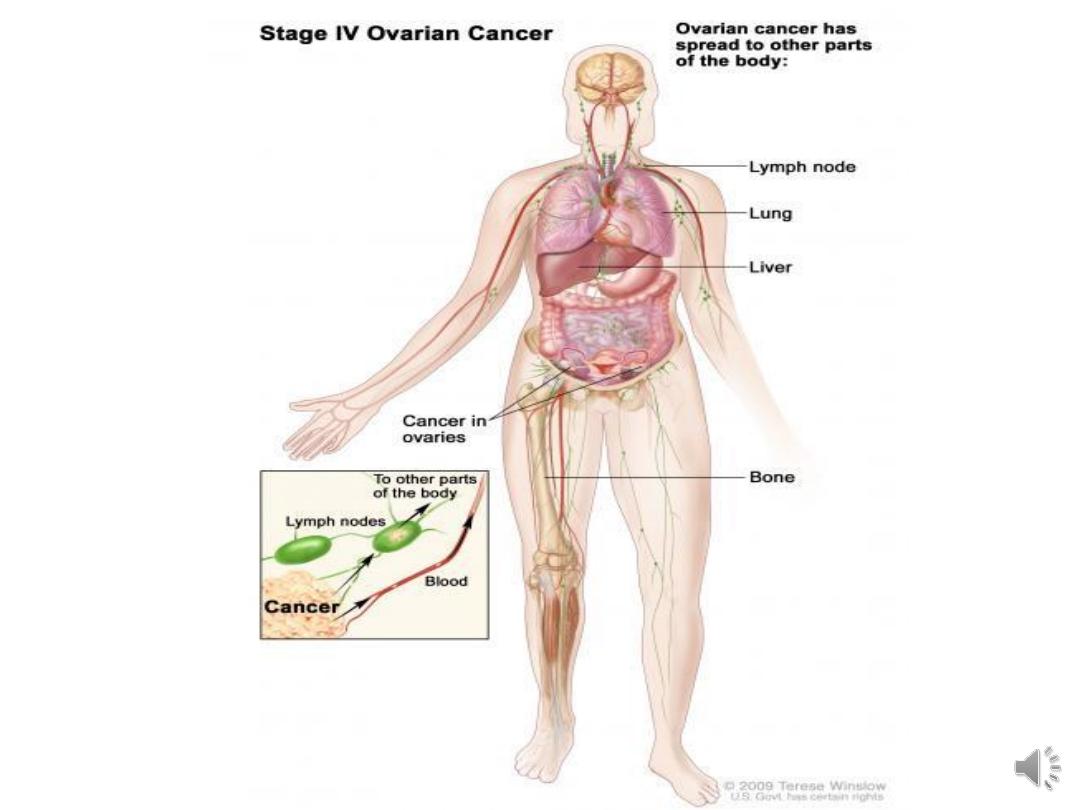
• Haematogenous spread occurs late in the
course of disease involving liver & lung,
sometimes bone & brain.

Clinical staging:
Stage FIGO definition
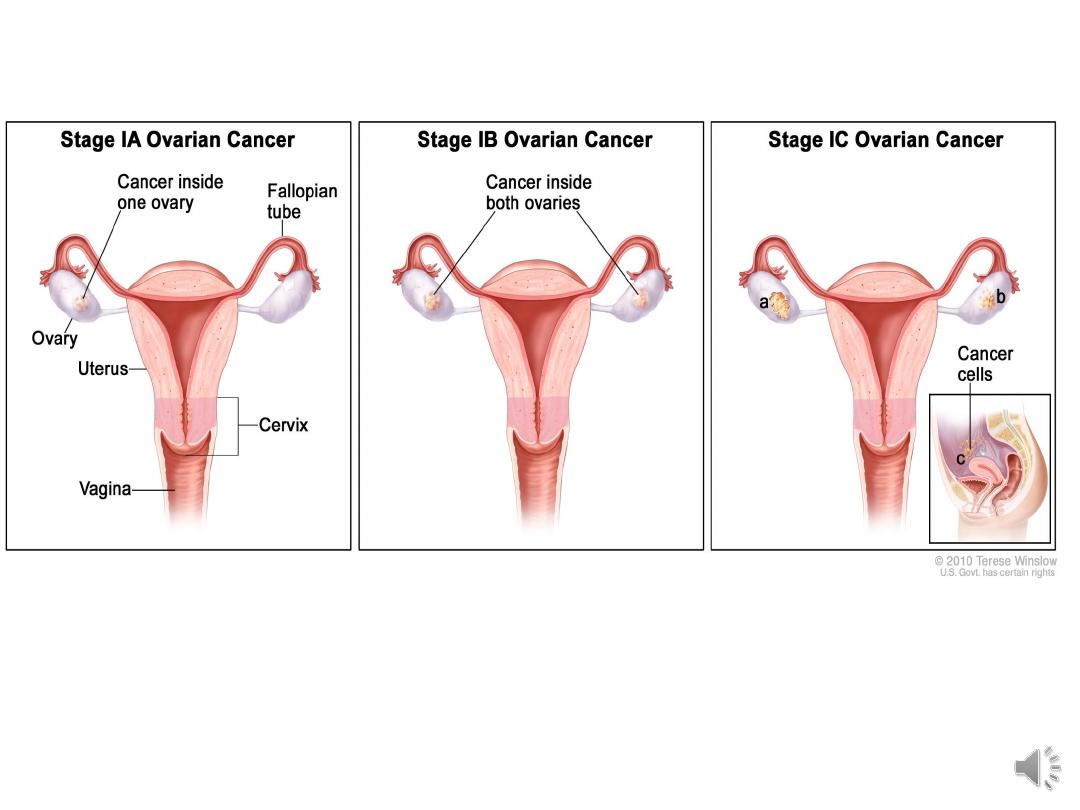
I growth limited to ovaries
Ia growth limited to one ovary, no ascites, no
tumour on external surface, capsule intact.
Ib growth limited to both ovaries, no ascites, no
tumour on external surfaces, capsule intact
Ic tumour either stage Ia or Ib but tumour on
surface of one or both ovaries or with capsule
rupture or with ascites present containing
malignant cells or with positive peritoneal
washing

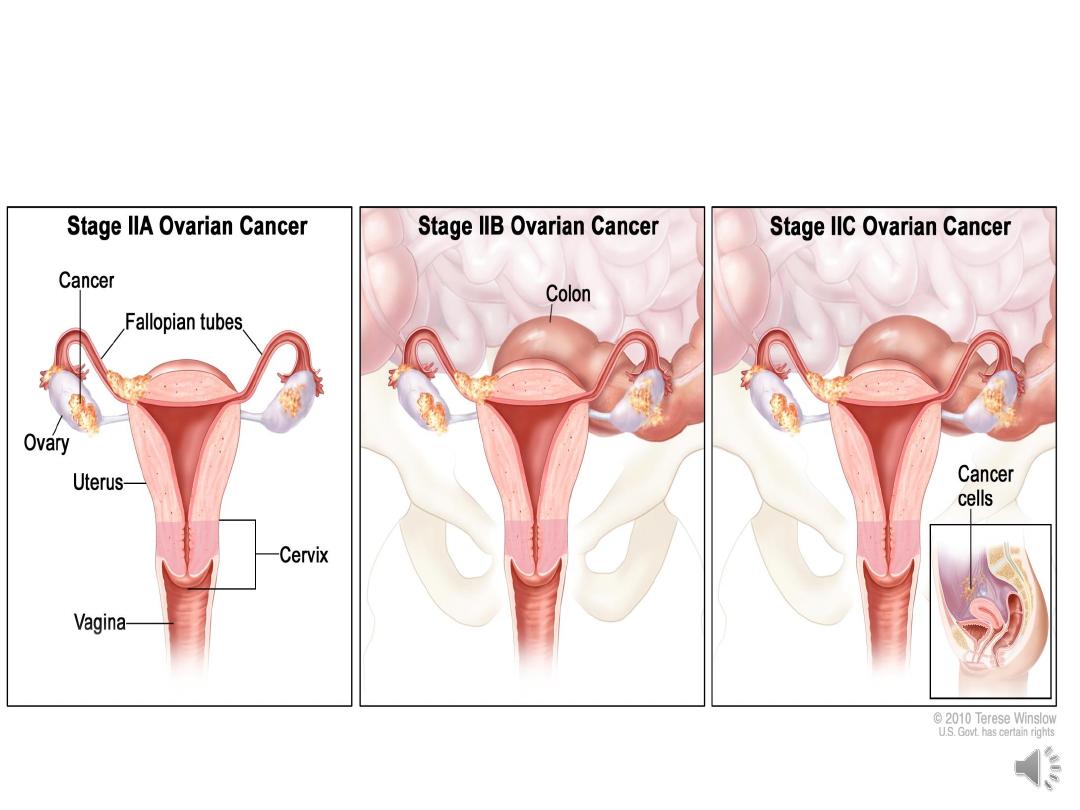
II growth involving one or both ovaries with
pelvic extension
IIa extension &/or metastasis to the uterus or
tubes
IIb extension to other pelvic tissues
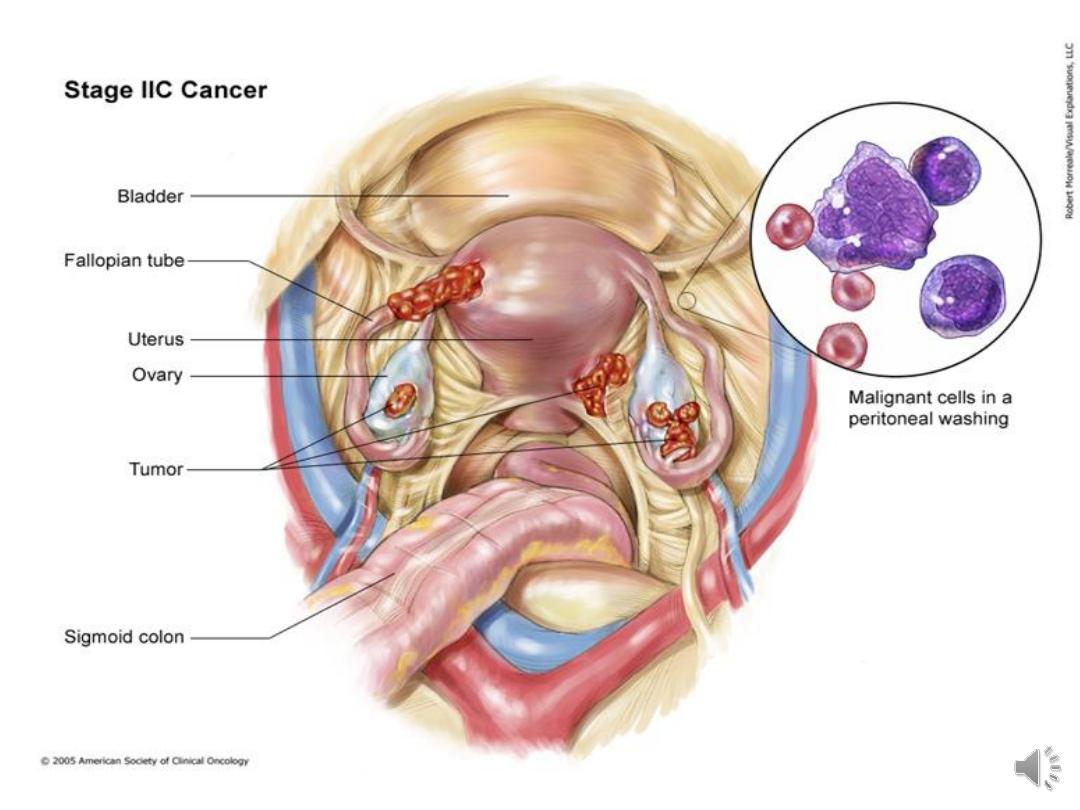
IIc tumour either stage IIa or IIb but tumour on
surface of one or both ovaries or with capsule
rupture or with ascites containing malignant
cells or with positive peritoneal washing

III tumour confined to abdominal peritonium or
positive retroperitoneal or inguinal nodes
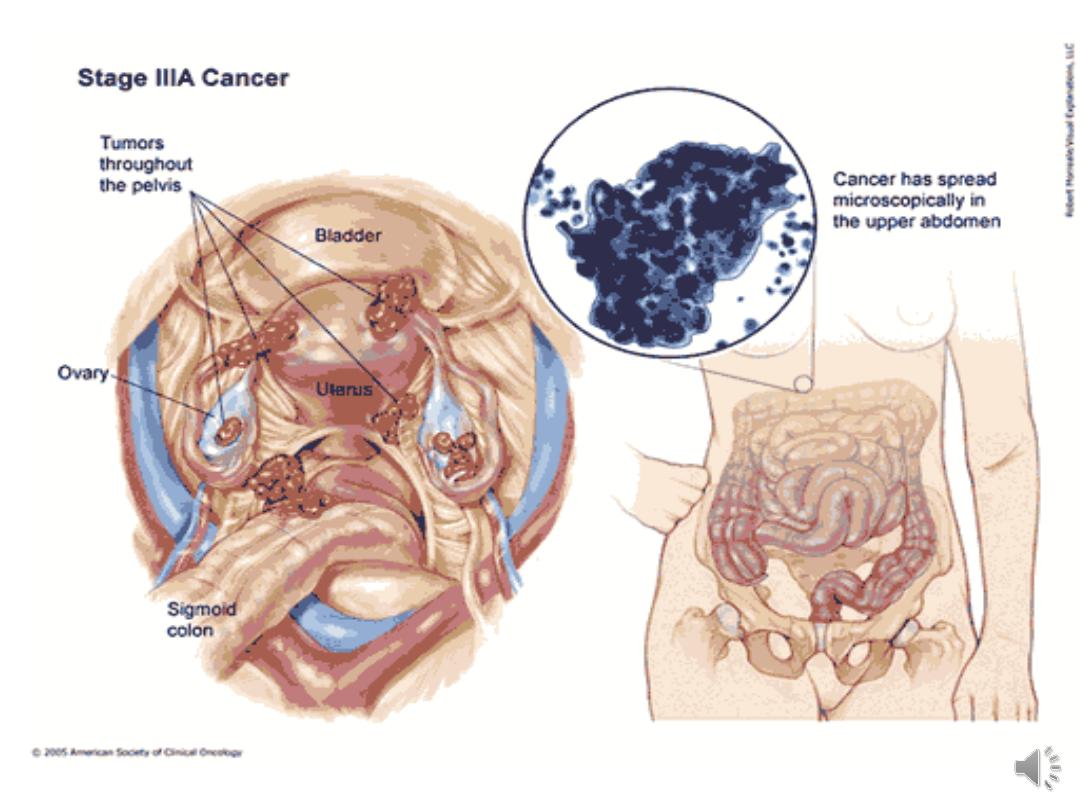
IIIa tumour grossly limited to the true pelvis with
negative nodes but with histologically confirmed
microscopic seeding of abdominal peritoneal surfaces
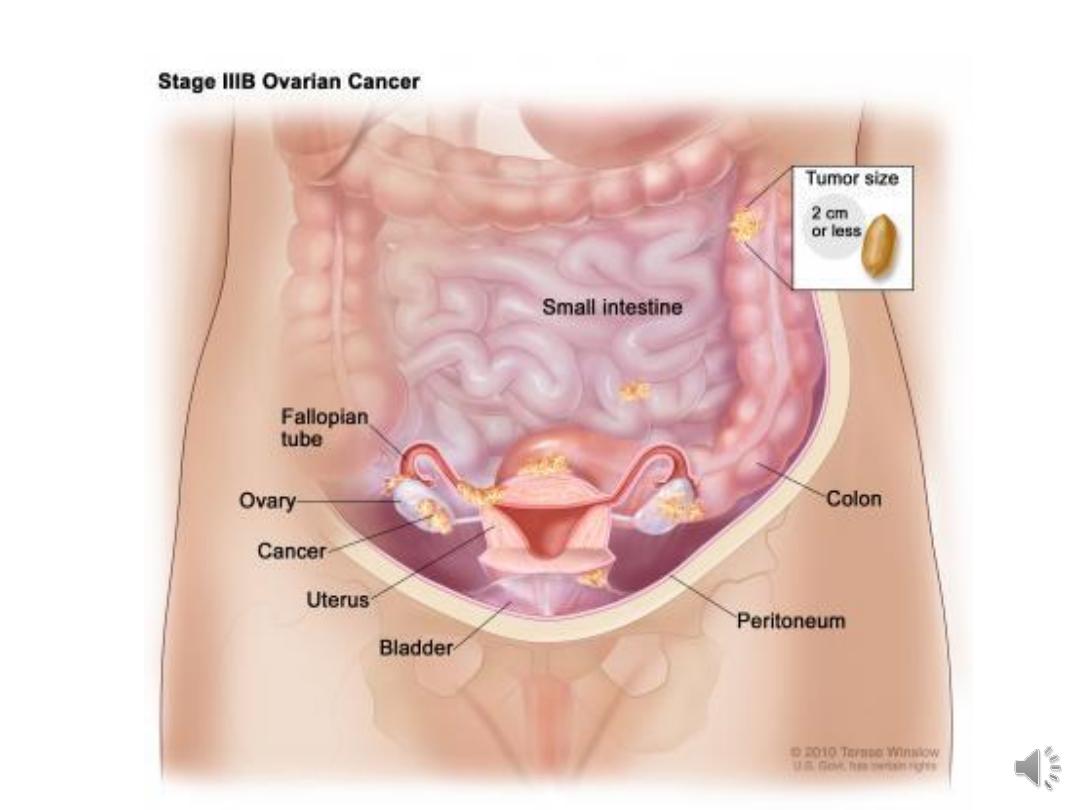
IIIb Abdominal implants <2 cm in diameter, nodes are
negative.
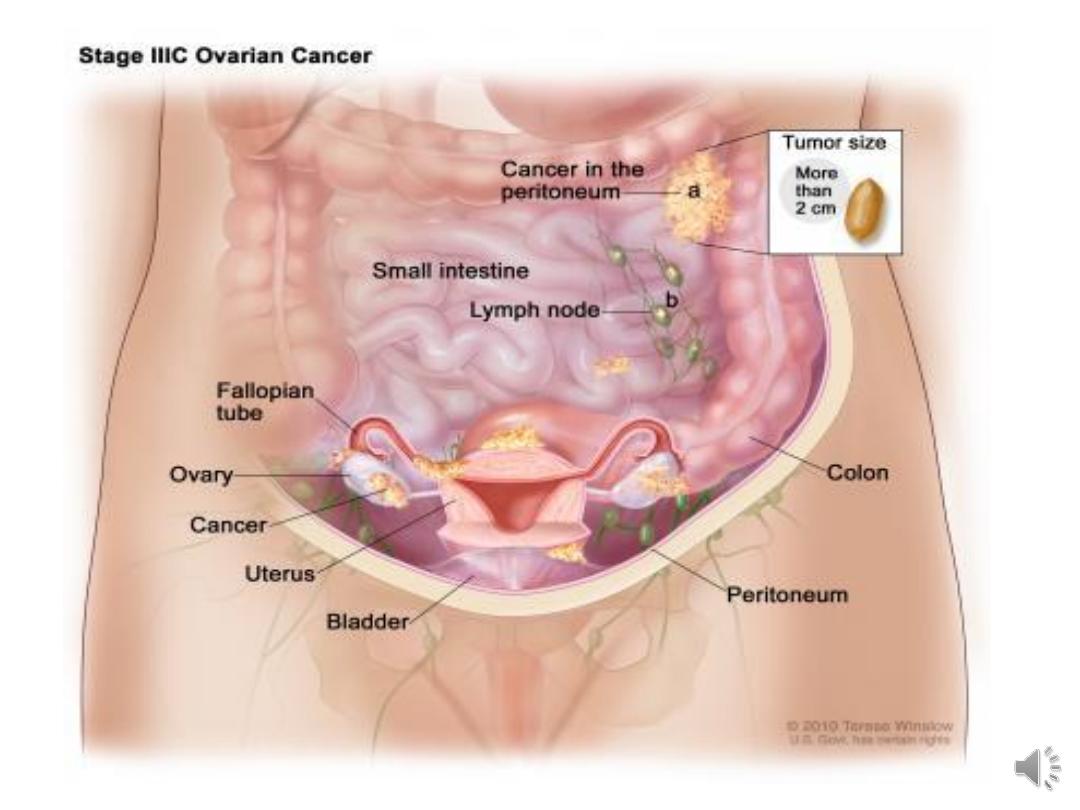
IIIc abdominal implants > 2cm in diameter or positive
retroperitoneal or inguinal nodes.
IV distant metastasis, if pleural effusion is present,
there must be positive cytology to allot a case to stage
IV
Surgery:
• surgery remains necessary for diagnosis, staging and
treatment of epithelial ovarian cancer.
• Surgery includes total hysterectomy, bilateral
salpingo-oophorectomy & infracolic omentectomy.
• The aim of surgery is complete or optimal
cytoreduction (where <1 cm of residual macroscopic
disease is left behind).

• In young nulliparous woman with unilateral
tumour & no ascites, unilateral salpigo-
oophorectomy may be justified after exploration
to exclude metastatic disease & curettage of the
uterine cavity to exclude synchronous
endometrial tumour.
• If the tumour is subsequently found to be poorly
differentiated or the washing is positive, a second
operation to clear the pelvis will be necessary.

• Interval debulking surgery: When bulky disease remains
after initial surgery,
chemotherapy
should be given in two
to four courses then a
second laparotomy
is performed
after which chemotherapy is resumed as soon as possible.
• If a patient is unfit or unwilling to have surgery, or if
preoperative assessment indicates that complete debulking
is unlikely to be achievable
, primary chemotherapy
may
be offered. If the patient responds to the chemotherapy,
interval surgery can be carried out after three cycles.
• In borderline tumours ovarian cystectomy or
oophorectomy are adequate in young women while
hysterectomy & bilateral salpingo-oophorectomy is
advisable for older women.

Chemotherapy:
• stage II-IV & stage Ic.
• given as primary treatment, as an adjunct
following surgery or for relapse of disease.
• given to prolong clinical remission & survival,
& for palliation in advanced & recurrent
disease.
• 3 weeks apart for six cycles.
• The platinum drugs, cisplatin & its analogue
carboplatin are heavy metal compounds which
cause cross-linkage of DNA strands.

• Carboplatin is the drug of choise, as effective as
cisplatin with lesser side effects.
• Paclitaxel works by causing microtubular damage to the
cell thus prevents replication and cell division.
• Bevacizumab, a monoclonal antibody against vascular
endothelial growth factor (VEGF), inhibits angiogenesis,
clinically effective at improving recurrence free and
overall survival when given in combination with
carboplatin and paclitaxel in advanced ovarian cancer
• Follow-up of patients includes clinical examination and
CA125 measurement
Prognosis: The overall 5-year survival from ovarian
cancer is 46%
Prognostic factors
Stage of diseae
Volume of residual disease post surgery
Histological type and grade of tumour
Age at presentation
FIGO stage
5-year survival (%)
1
80–90%
2
65–70%
3
30–50%
4
15%

Primary peritoneal carcinoma
• PPC is a high-grade pelvic serous carcinoma, histologically
indistinct from tumours arising from the Fallopian tube or
ovary.
• Criteria for diagnosis includes:
– Normal sized or slightly bulky ovaries.
– More extraovarian disease than ovarian disease.
– Low volume peritoneal disease.
• The clinical behaviour, prognosis and treatment is the same as
for other high-grade pelvic serous carcinomas, with a trend
towards using primary chemotherapy as complete surgical
debulking is difficult.

Sex cord stromal tumours:
• they are tumours of low malignant potential
with a good long-term prognosis.
• morbidity may arise from the oestrogen
(granulosa, theca cell) or androgen production
(Seroli–Leydig)
• Presentation
• staging system is the same as for epithelial
tumours. Most present as stage I.
• Treatment: surgical treatment is the same as
for epithelial tumours. Unilateral
oophorectomy is indicated in young women
with stage Ia disease.

Malignant Germ cell tumours: mainly in young women.
• Dysgerminomas account for 50% of all germ cell tumours.
occasionally secrete human chorionic gonadotrophin
(hCG).
• Endodermal sinus yolk sac tumours are the second most
common germ cell tumours, accounting for 15% of the
total. secrete α-fetoprotein (AFP). present with a large
solid mass that often causes acute symptoms with torsion
or rupture.
• Immature teratomas account for 15–20% of malignant
germ cell tumours
• Non-gestational choriocarcinomas are very rare, usually
presenting in young girls with irregular bleeding and very
high levels of hCG.

Clinical features:
• Present in young woman with a large solid ovarian
mass that is rapidly growing.
• Tumour markers are measured preoperatively.
• MRI is helpful to assess morphology, particularly
within teratomas.
• CT scaning of the abdomen allows assessment of the
liver and lymph nodes.
• All patients should have a chest X-ray to exclude
pulmonary metastases

Treatment:
• fertility-sparing treatment may be preferred for young
patients.
• exploratory laparotomy, remove the tumour and assess
contralateral ovary. Careful inspection of the abdominal
cavity, peritoneal biopsies and sampling of any enlarged
pelvic or para-aortic nodes.
• Postoperative chemotherapy depends on stage of disease.
combination of bleomycin, etoposide and cisplatin (BEP),
given as a course of three to four treatments, 3 weeks
apart. This regime gives long-term cure rates of over 90%
and also preserves fertility if required.

