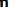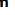The Urinary System
Prof.dr.maha shakir alyasiriIntroduction
The urinary system consists of:paired kidneys
ureters,
bladder
urethra.
This system’s primary role is to ensure optimal properties of the blood, which the kidneys continuously monitor.
Regulation of the balance between water and electrolytes (inorganic ions) and the acid-base balance;
Excretion of metabolic wastes along with excess water and electrolytes in urine, the kidneys’ excretory product which passes through the ureters for temporary storage in the bladder before its release to the exterior by the urethra;
Renal functions:
Excretion of many bioactive substances, including many drugs;Secretion of renin, a protease important for regulation of blood pressure by cleaving circulating angiotensinogen to angiotensin I;
Secretion of erythropoietin, a glycoprotein growth factor that stimulates erythrocyte production in red marrow when the blood O2 level is low;
Conversion of the steroid prohormone vitamin D, initially produced in the skin, to the active form (1,25-dihydroxyvitamin D3 or calcitriol); and
Gluconeogenesis during starvation or periods of prolonged fasting, making glucose from amino acids to supplement this process in the liver.
KIDNEYS
Approximately 12-cm long, 6-cm wide, and 2.5-cm thick in adults,
each kidney has a concave medial border, the hilum where nerves enter, the ureter exits, and blood and lymph vessels enter and exit
and a convex lateral surface,
both covered by a thin fibrous capsule.
Within the hilum the upper end of the ureter expands as the renal pelvis and
renal pelvis divides into two or three major calyces.
Smaller branches, the minor calyces, arise from each major calyx.
The parenchyma of each kidney has an outer renal cortex, a darker stained region with many round corpuscles and tubule cross sections,
and an inner renal medulla consisting mostly of aligned linear tubules and ducts.
The renal medulla in humans consists of 8-15 conical structures called renal pyramids, all with their bases meeting the cortex (at the corticomedullary junction) and separated from each other by extensions of the cortex called renal columns.
Each pyramid plus the cortical tissue at its base and extending along its sides constitutes a renal lobe.
The tip of each pyramid, called the renal papilla, projects into a minor calyx that collects urine formed by tubules in one renal lobe
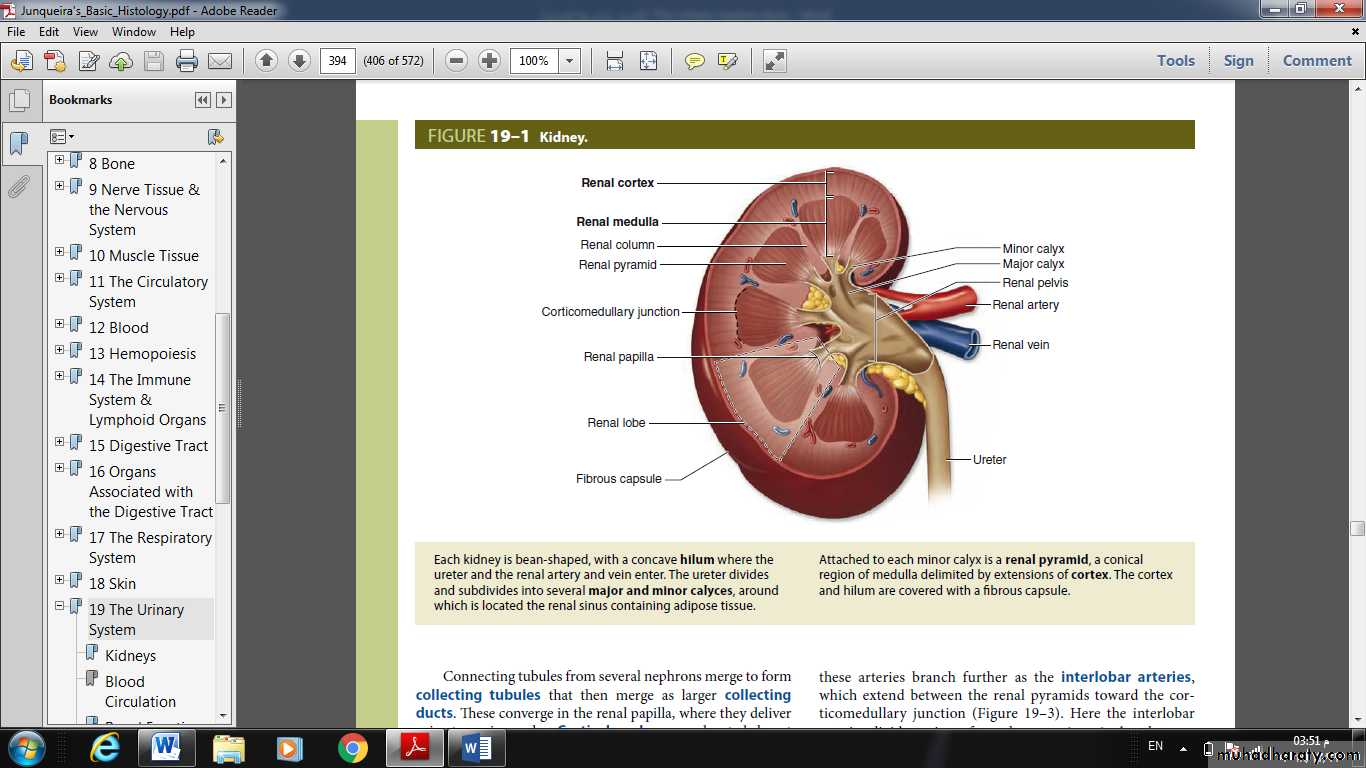
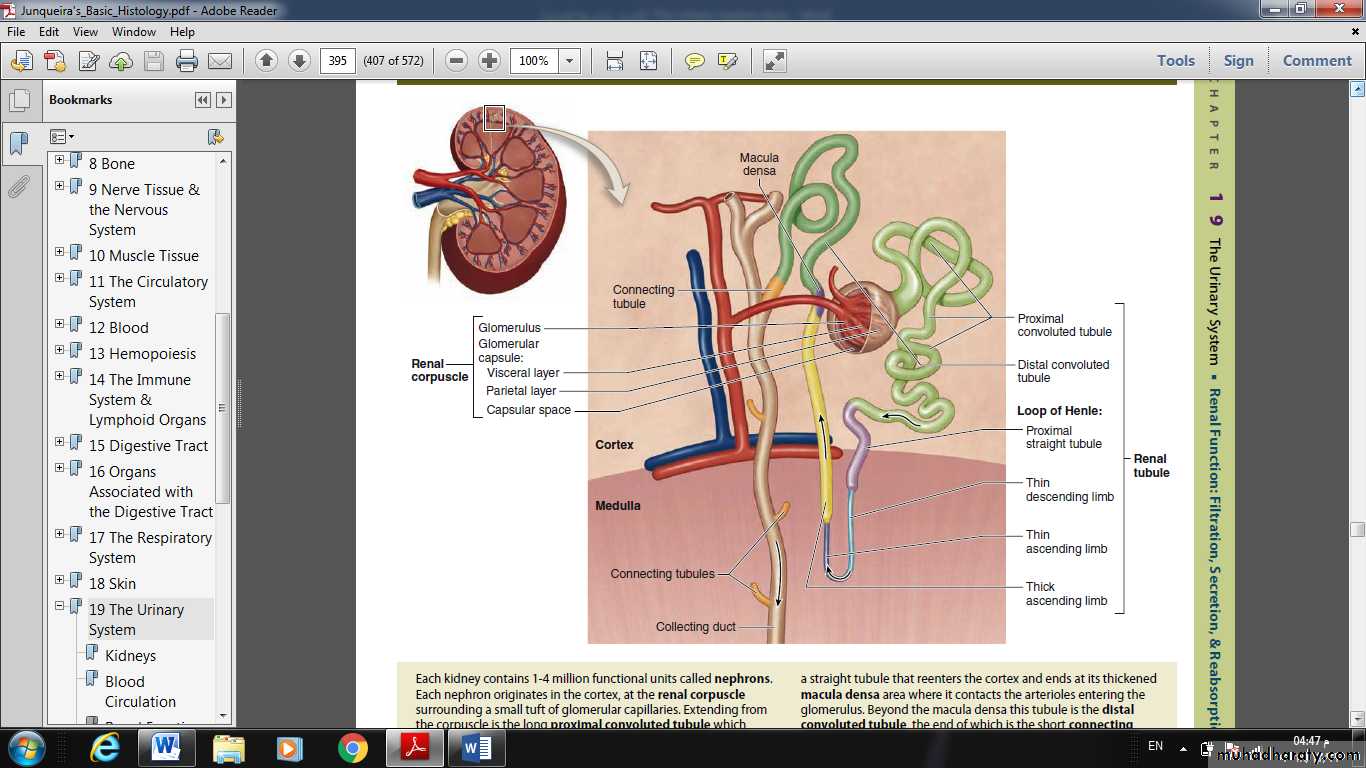
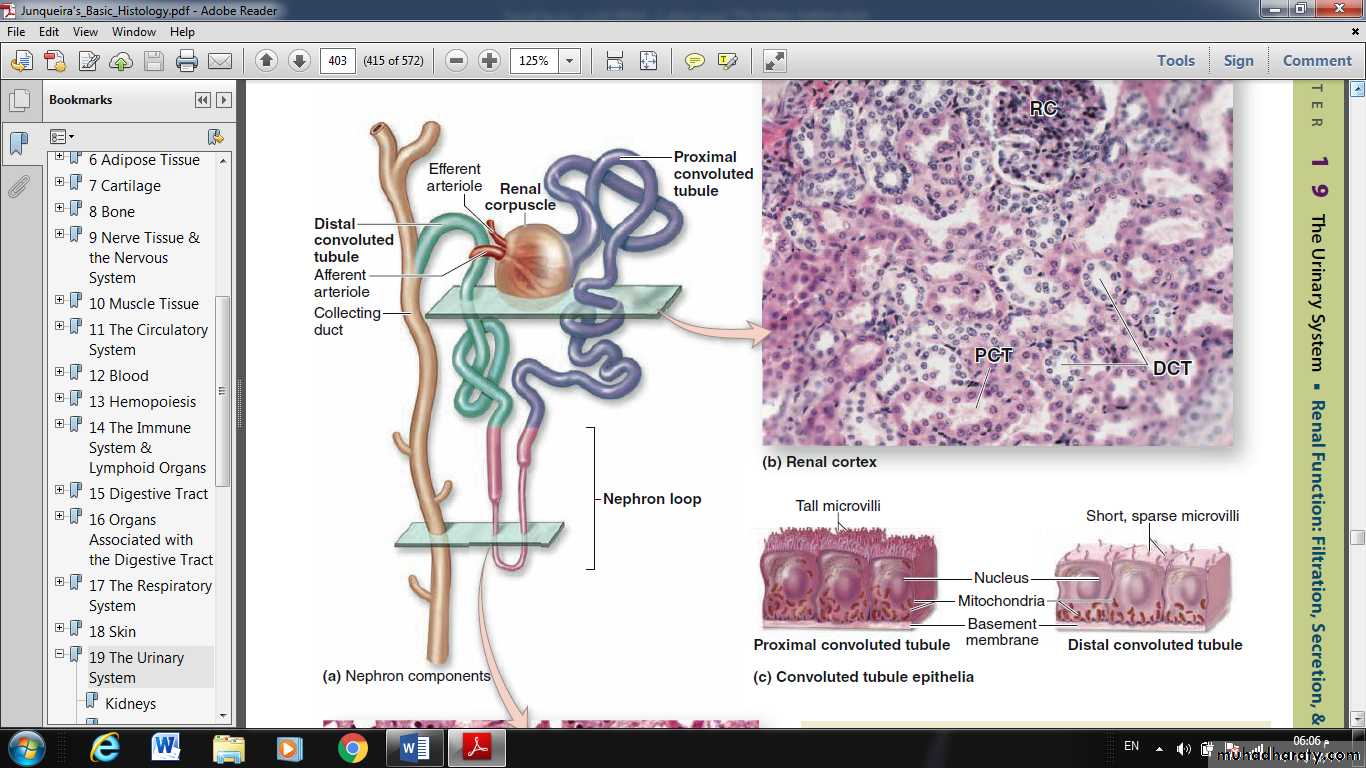
Kidneys each contain 1-4 million functional units called nephrons , each consisting of a corpuscle and a long, simple epithelial renal tubule with three main parts along its length.
The major divisions of each nephron are:
■ Renal corpuscle, an initial dilated part enclosing a tuft of capillary loops and the site of blood filtration, always located in the cortex;■ Proximal tubule, a long convoluted part, located entirely in the cortex, with a shorter straight part that enters the medulla;
• ■ Loop of Henle (or nephron loop), in the medulla, with a thin descending and a thin ascending limb;
• ■ Distal tubule, consisting of a thick straight part ascending from the loop of Henle back into the cortex and a convoluted part completely in the cortex;
Connecting tubule, a short minor part linking the nephron to collecting ducts.
• Connecting tubules from several nephrons merge to form collecting tubules that then merge as larger collecting ducts. These converge in the renal papilla, where they deliver urine to a minor calyx.• Cortical nephrons are located almost completely in the cortex while juxtamedullary nephrons (about one-seventh of the total) lie close to the medulla and have long loops of Henle.
BLOOD CIRCULATION
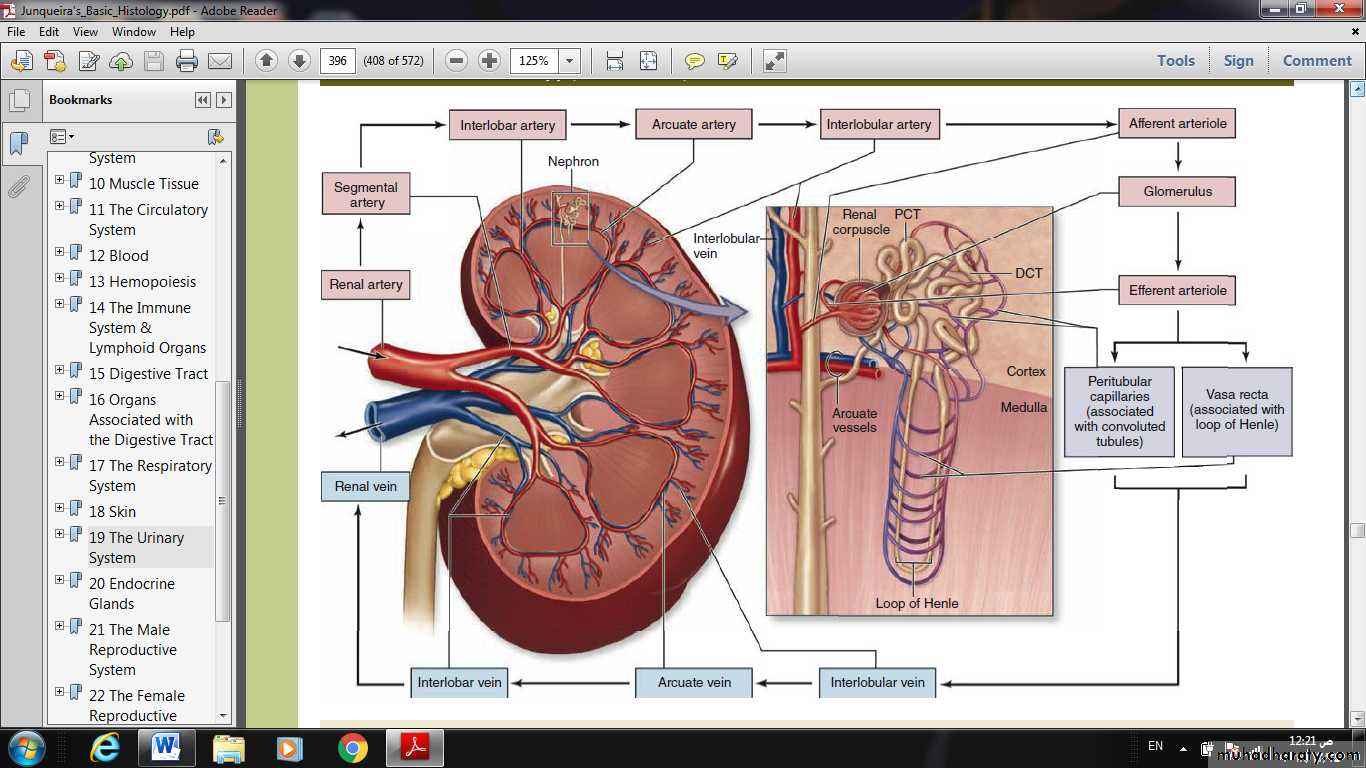
• Each kidney’s renal artery divides into two or more segmental arteries at the hilum.• Around the renal pelvis, these arteries branch further as the interlobar arteries, which extend between the renal pyramids toward the corticomedullary junction.
• Here the interlobar arteries divide again to form the arcuate arteries that run in an arc along this junction at the base of each renal pyramid.
• Smaller interlobular arteries radiate from the arcuate arteries, extending deeply into the cortex.
• From the interlobular arteries arise the microvascular afferent arterioles, which divide to form a plexus of capillary loops called the glomerulus, each of which is located within a renal corpuscle where the blood is filtered.
• Blood leaves the glomerular capillaries, via efferent arterioles, which branch again to form another capillary network, usually the peritubular capillaries profusely distributed throughout the cortex. From the juxtaglomerular corpuscles near the medulla, efferent arteriole branch repeatedly to form parallel bundles of capillary loops called the vasa recta that penetrate deep into the medulla in association with the loops of Henle and collecting ducts.
Collectively, the cortex receives over 10 times more blood than the medulla.
Blood leaves the kidney in veins that follow the same courses as arteries and have the same names.
RENAL FUNCTION: FILTRATION, SECRETION, & REABSORPTION
All the major functions of the kidneys the removal of metabolic wastes and excess water and electrolytes from blood are performed by various specialized epithelial cells of the nephrons and collecting systems.Renal function involves specific activities:
■ Filtration, by which water and solutes in the blood leave the vascular space and enter the lumen of the nephron■Tubular secretion, by which substances move from epithelial cells of the tubules into the lumens, usually after uptake from the surrounding interstitium and capillaries
■Tubular reabsorption
• by which substances move from the tubular lumen across the epithelium into the interstitium and surrounding capillaries. Along the length of the nephron tubule and collecting system,
• the filtrate receives various secreted molecules while others are reabsorbed and then enters the minor calyces as urine and undergoes excretion.
• The number of nephrons decreases slightly in older adults,
• a process accelerated by high blood pressure.• If a kidney is donated for transplant (unilateral nephrectomy), the remaining kidney undergoes compensatory growth with cellular hypertrophy in the proximal parts of the nephron tubules and an increase in the rate of filtration, which allow normal renal function to continue.
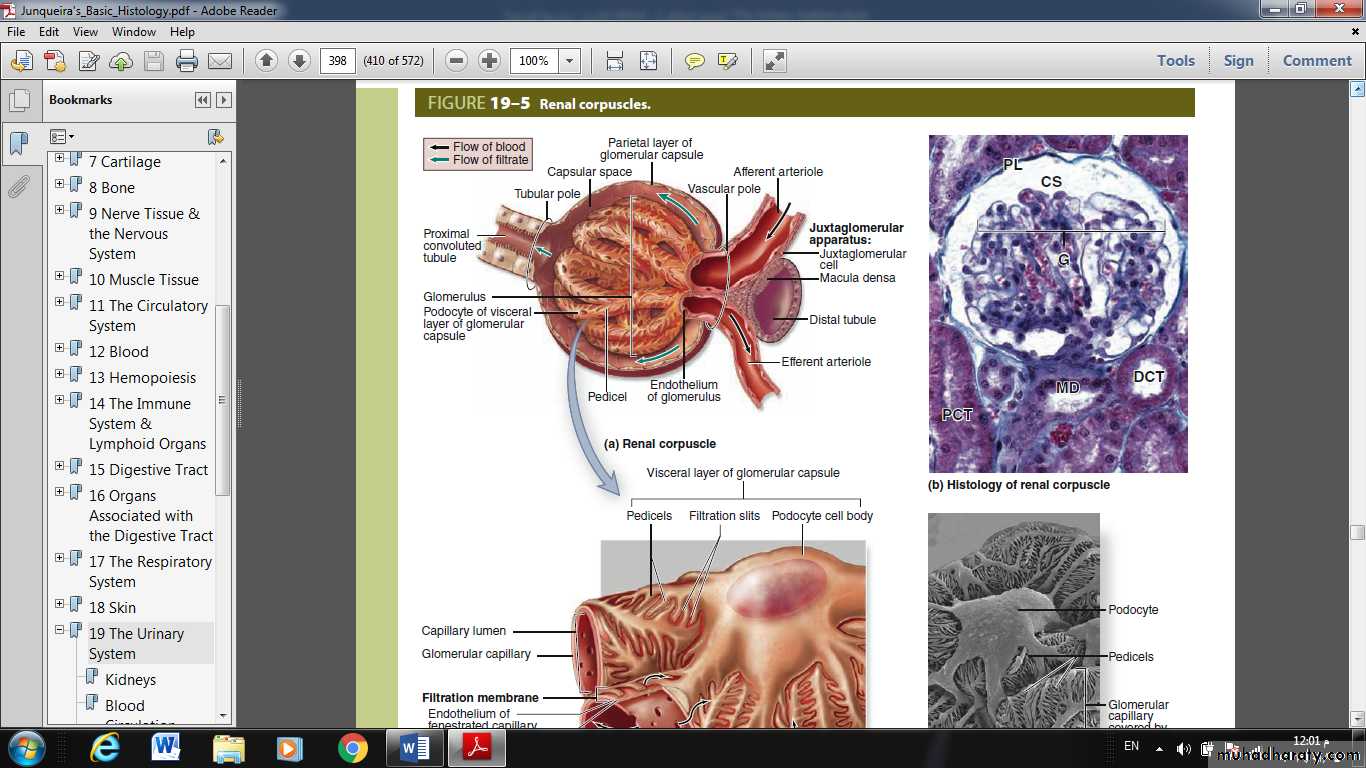
Renal Corpuscles & Blood Filtration
• At the beginning of each nephron is a renal corpuscle, about 200 μm in diameter and containing a tuft of glomerular capillaries, surrounded by a double-walled epithelial capsule called the glomerular (Bowman) capsule.The internal or visceral layer of this capsule closely envelops the glomerular capillaries, which are finely fenestrated.
The outer parietal layer forms the surface of the capsule. Between the two capsular layers is the capsular (or urinary) space, which receives the fluid filtered through the capillary wall and visceral layer.
Each renal corpuscle has a vascular pole, where the afferent arteriole enters and the efferent arteriole leaves, and a tubular pole, where the proximal convoluted tubule (PCT) begins.
The outer parietal layer of a glomerular capsule consists of a simple squamous epithelium supported externally by a basal lamina. At the tubular pole, this epithelium changes to the simple cuboidal epithelium that continues and forms the proximal tubule.
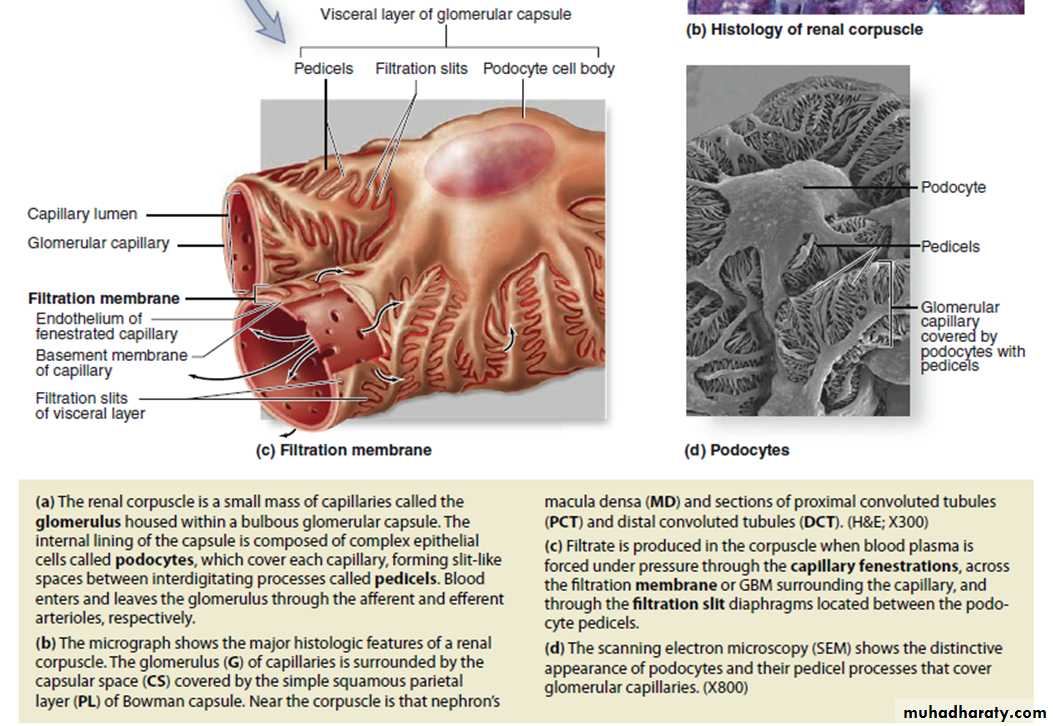
The visceral layer of a renal corpuscle consists of unusual stellate epithelial cells called podocytes, which together with the capillary endothelial cells compose the apparatus for renal filtration.
From the cell body of each podocyte several primary processes extend and curve around a length of glomerular capillary.
Each primary process gives rise to many parallel, interdigitating secondary processes or pedicels (L. pedicellus, little foot;). The pedicels cover much of the capillary surface, in direct contact with the basal lamina.
Between the interdigitating pedicels are elongated spaces, or filtration slit pores.
Slit diaphragms are modified and specialized occluding or tight junctions composed of nephrins, other proteins, glycoproteins, and proteoglycans important for renal function.
Between the highly fenestrated endothelial cells of the capillaries and the covering podocytes is the thick glomerular basement membrane (GBM).
This membrane is the most substantial part of the filtration barrier that separates the blood from the capsular space and forms by fusion of the capillary- and podocyte-produced basal laminae, restricts passage of proteins larger than about 70 kDa.
Smaller proteins that are filtered from plasma are degraded, and the amino acids reabsorbed in the proximal tubule.
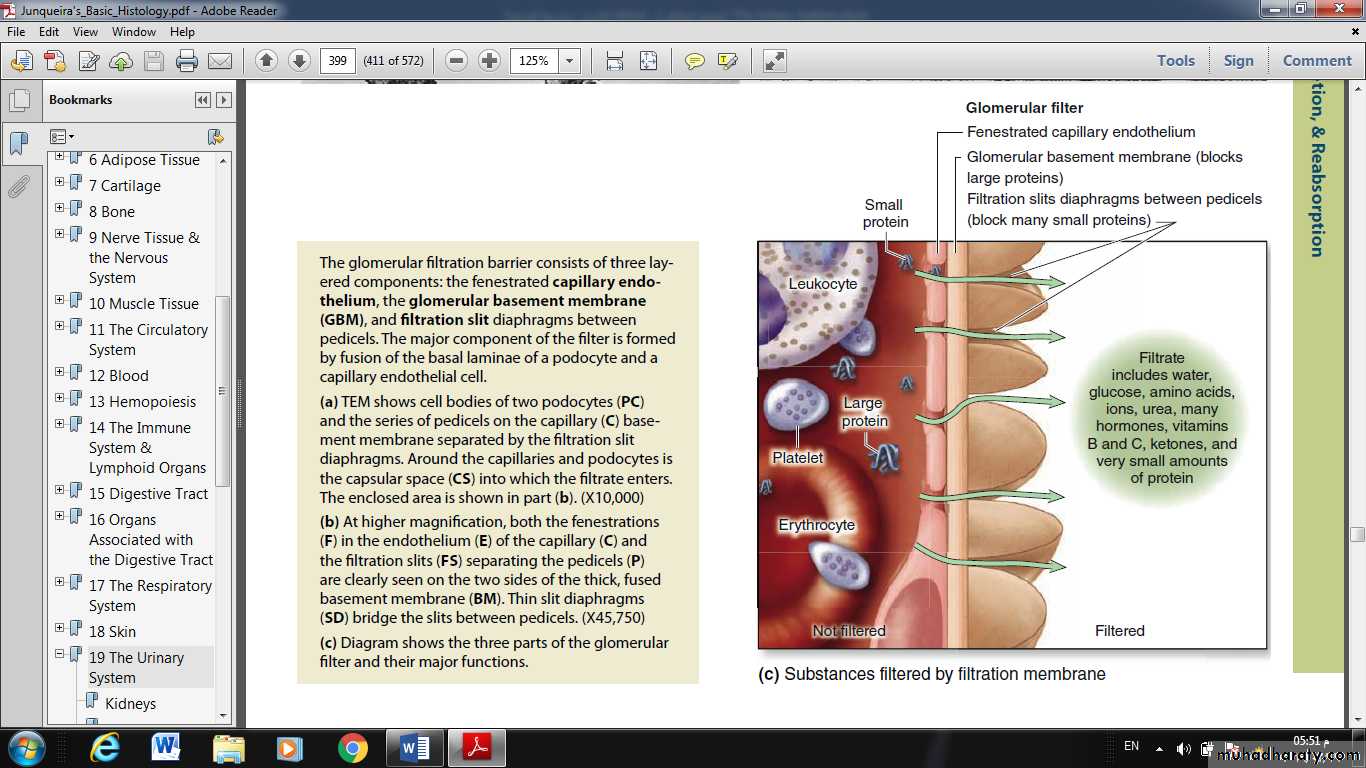
Filtration, therefore, occurs through a structure with three parts:
• The fenestrations of the capillary endothelium, which blocks blood cells and platelets
• GBM, which restricts large proteins and some organic anions
• The filtration slit diaphragms between pedicels, which restrict some small proteins and organic anions.
• Normally about 20% of the blood plasma entering a glomerulus is filtered into the capsular space.
In diseases such as diabetes mellitus and glomerulonephritis, the release of protein into the urine (proteinuria).
The total glomerular filtration area of an adult 500 cm2 and the average GFR 180 L/d.
Because the total amount of circulating plasma averages 3 L, it follows that the kidneys typically filter the entire blood volume 60 times every day.
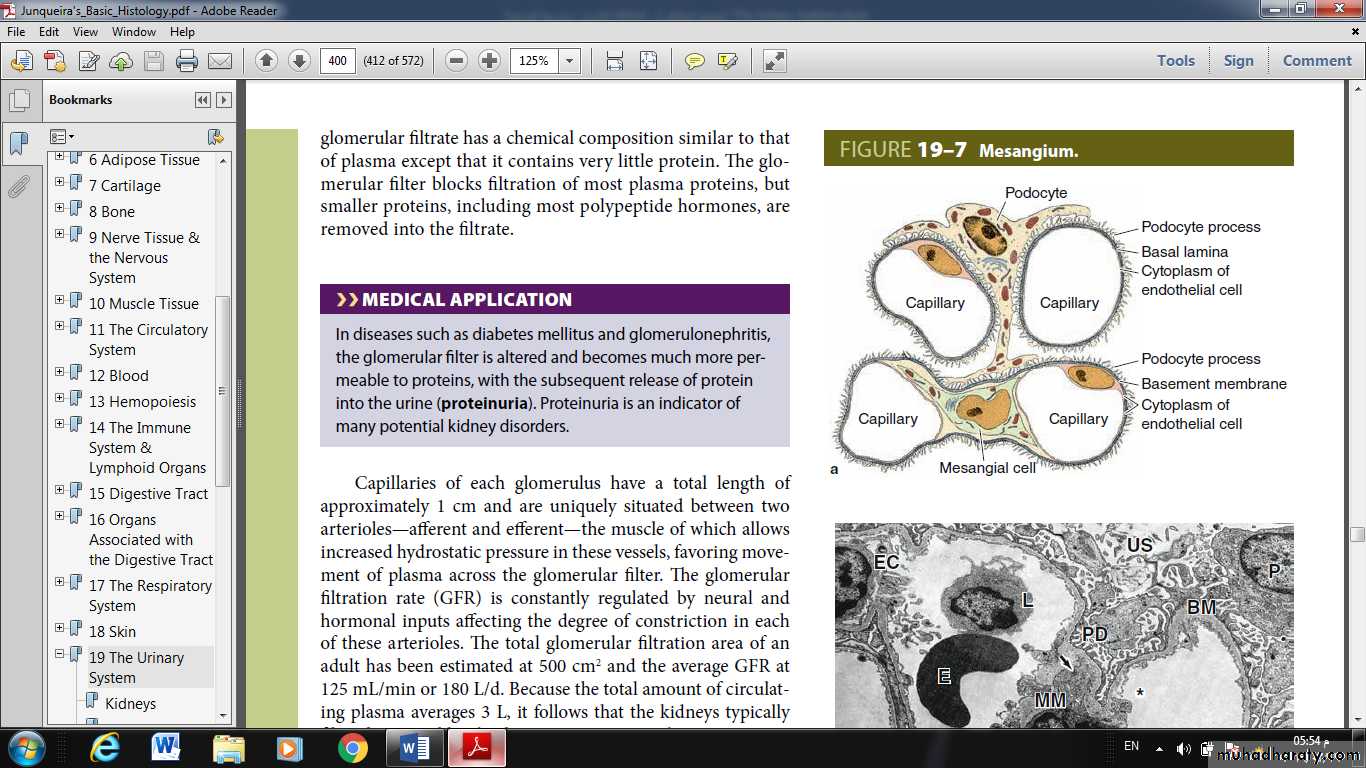
In addition to capillary endothelial cells and podocytes, renal corpuscles also contain mesangial cells (Gr. mesos, in the midst + angion, vessel), most of which resemble vascular pericytes in having contractile properties and producing components of an external lamina. They and their surrounding matrix comprise the mesangium, which fills interstices between capillaries.
Functions of the mesangium include the following:
• Physical support of capillaries within the glomerulus
• Adjusted contractions in response to blood pressure changes, which help maintain an optimal filtration rate
• Phagocytosis of protein aggregates adhering to the glomerular filter,
• Secretion of several cytokines, prostaglandins, and other factors important for immune defense and repair
At the tubular pole of the renal corpuscle, the simple squamous epithelium of the capsule’s parietal layer is continuous with the simple cuboidal epithelium of the proximal convoluted tubule (PCT) These long, tortuous tubules fill most of the cortex. PCT cells are specialized.
Proximal Convoluted Tubule
Cells in many parts of the nephron tubule and collecting system reabsorb water and electrolytes, but other activities are restricted mainly to specific tubular regions.At the tubular pole of the renal corpuscle, the simple squamous epithelium of the capsule’s parietal layer is continuous with the simple cuboidal epithelium of the proximal convoluted tubule (PCT) These long, tortuous tubules fill most of the cortex.
PCT cells are specialized for both reabsorption and secretion.
Over half of the water and electrolytes, and all of the organic nutrients (glucose, amino acids, vitamins, etc.), filtered from plasma in the renal corpuscle are normally reabsorbed in the PCT.
These molecules are transferred directly across the tubular wall for immediate uptake again into the plasma of the peritubular capillaries.
Conversely, organic anions and cations not filtered in the renal corpuscle may be released in the peritubular capillaries, taken up by the cells of the proximal tubules and undergo secretion into the filtrate.
Organic anion and cation transporters allow the kidneys to dispose of such substances at a higher rate than by glomerular filtration alone.
Because these molecules include important substances (such as bile salts, creatinine, etc.) and many antibiotics and other drugs, this process of tubular secretion is of great pharmacologic importance as a key mechanism of drug clearance.
The cells of PT have:
central nuclei and
very acidophilic cytoplasm because of the abundant mitochondria.
The cell apex has very many long microvilli that form a prominent brush border in the lumen that facilitates reabsorption.
Because the cells are large, each transverse section of a PCT typically contains only three to five nuclei.
Peritubular capillaries are abundant in the sparse surrounding connective tissue interstitium, which fills only about 10% of the cortex.
fibroblastic interstitial cells in cortical areas near the proximal tubules produce erythropoietin. Besides their major roles in reabsorption and secretion.
cells of the proximal tubule also perform hydroxylation of vitamin D and release to the capillaries.
• Loop of Henle
The PCT continues with the much shorter proximal straight tubule that enters the medulla and continues as the nephron’s loop of Henle.
This is a U-shaped structure with a thin descending limb and a thin ascending limb, both composed of simple squamous epithelia.
The straight part of the proximal tubule has an outer diameter of about 60 μm, but it narrows abruptly to about 30 μm in the thin limbs of the loop.
The wall of the thin segments consists only of squamous cells with few organelles (indicating a primarily passive role in transport) and the lumen is prominent.
The thin ascending limb of the loop becomes the thick ascending limb (TAL), with simple cuboidal epithelium and many mitochondria again, in the outer medulla and extends as far as the macula densa near the nephron’s glomerulus.
The loops of Henle and surrounding interstitial connective tissue are involved in further adjusting the salt content of the filtrate.
Cuboidal cells of the loops’ TALs actively transport sodium and chloride ions out of the tubule against a concentration gradient into the hyaluronate-rich interstitium, making that compartment hyperosmotic. This causes water to be withdrawn passively from the thin descending part of the loop, thus concentrating the filtrate.
The thin ascending limbs reabsorb sodium chloride (NaCl) but are impermeable to water.
The countercurrent flow of the filtrate (descending, then immediately ascending) in the two parallel thin limbs establishes a gradient of osmolarity in the interstitium of the renal pyramids, an effect that is “multiplied” at deeper levels in the medulla.
The interstitial osmolarity at the pyramid tips is about four times that of the blood.
Distal Convoluted Tubule & Juxtaglomerular Apparatus
The ascending limb of the nephron is straight as it enters the cortex and forms the macula densa, and then becomes tortuous as the distal convoluted tubule (DCT).Much less tubular reabsorption occurs here than in the proximal tubule. The simple cuboidal cells of the distal tubules differ from those of the proximal tubules in being smaller and having no brush border and more empty lumens.
Because distal tubule cells are flatter and smaller than those of the proximal tubule, more nuclei are typically seen in sections of distal tubules than in those of proximal tubules.
Cells of the DCT also have fewer mitochondria than cells of proximal tubules, making them less acidophilic.
The rate of Na+ absorption here is regulated by aldosterone from the adrenal glands.
Where the initial, straight part of the distal tubule contacts the arterioles at the vascular pole of the renal corpuscle of its parent nephron, its cells become more columnar and closely packed, forming the macula densa (L. thicker spot).
This is part of a specialized sensory structure, the juxtaglomerular apparatus (JGA) that utilizes feedback mechanisms to regulate glomerular blood flow and keep the rate of glomerular filtration relatively constant
Cells of the macula densa typically have apical nuclei, basal Golgi complexes, and a more elaborate and varied system of ion channels and transporters. Adjacent to the macula densa, the tunica media of the afferent arteriole is also modified. The smooth muscle cells are modified as juxtaglomerular granular (JG) cells, with a secretory phenotype including more rounded nuclei, rough ER, Golgi complexes, and granules with the protease renin.
Also at the vascular pole are lacis cells (Fr. lacis, lacework), which are extraglomerular mesangial cells that have many of the same supportive, contractile, and defensive functions as these cells inside the glomerulus.
Basic functions of the JGA in the autoregulation of the GFR and in controlling blood pressure include the following activities. Elevated arterial pressure increases glomerular capillary blood pressure, which increases the GFR. Higher GFR leads to higher luminal concentrations of Na+ and Cl– in the TAL of the nephron, which are monitored by cells of the macula densa.
Increased ion levels in the lumen cause these cells to release ATP, adenosine, and other vasoactive compoundsthat trigger contraction of the afferent arteriole, which lowers glomerular pressure and decreases the GFR. This lowers tubular ion concentrations, which turns off the release of vasoconstrictors from the macula densa.
Decreased arterial pressure leads to increased autonomic stimulation to the JGA as a result of baroreceptor function, including local baroreceptors in the afferent arteriole, possibly the JG cells themselves. This causes the JG cells to release renin, protease, into the blood.
There renin cleaves the plasma protein angiotensinogen into the inactive decapeptide angiotensin I. Angiotensin-converting enzyme (ACE) on lung capillaries clips this further to angiotensin II, a potent vasoconstrictor that directly raises systemic blood pressure and stimulates the adrenals to secrete aldosterone.
Aldosterone promotes Na+ and water reabsorption in the distal convoluted and connecting tubules, which raises blood volume to help increase blood pressure. The return of normal blood pressure turns off secretion of renin by JG cells.
Collecting Ducts
The last part of each nephron, the connecting tubule, carries the filtrate into a minor calyx.in which more water is reabsorbed if needed by the body
a connecting tubule extends from each nephron and several join together to form collecting ducts
simple cuboidal epithelium
an average diameter of 40 μm.
In the medulla these merge further, forming larger and straighter collecting ducts with increasingly columnar cells and overall diameters reaching 200 μ.
Approaching the apex of each renal pyramid, several medullary collecting ducts.
Antidiuretic hormone (ADH), released from the posterior pituitary gland as the body becomes dehydrated,
makes collecting ducts more permeable to water and increases the rate at which water molecules are pulled osmotically from the filtrate.
The water thus saved immediately enters the blood in the vasa recta.
Scattered among the principal cells are variably darker intercalated cells with more abundant mitochondria and projecting apical folds. Intercalated cells, some of which also occur in the DCTs, help maintain acid-base balance by secreting either H+ (from type A or α intercalated cells) or HCO3– (from type B or β intercalated cells).URETERS, BLADDER, & URETHRA
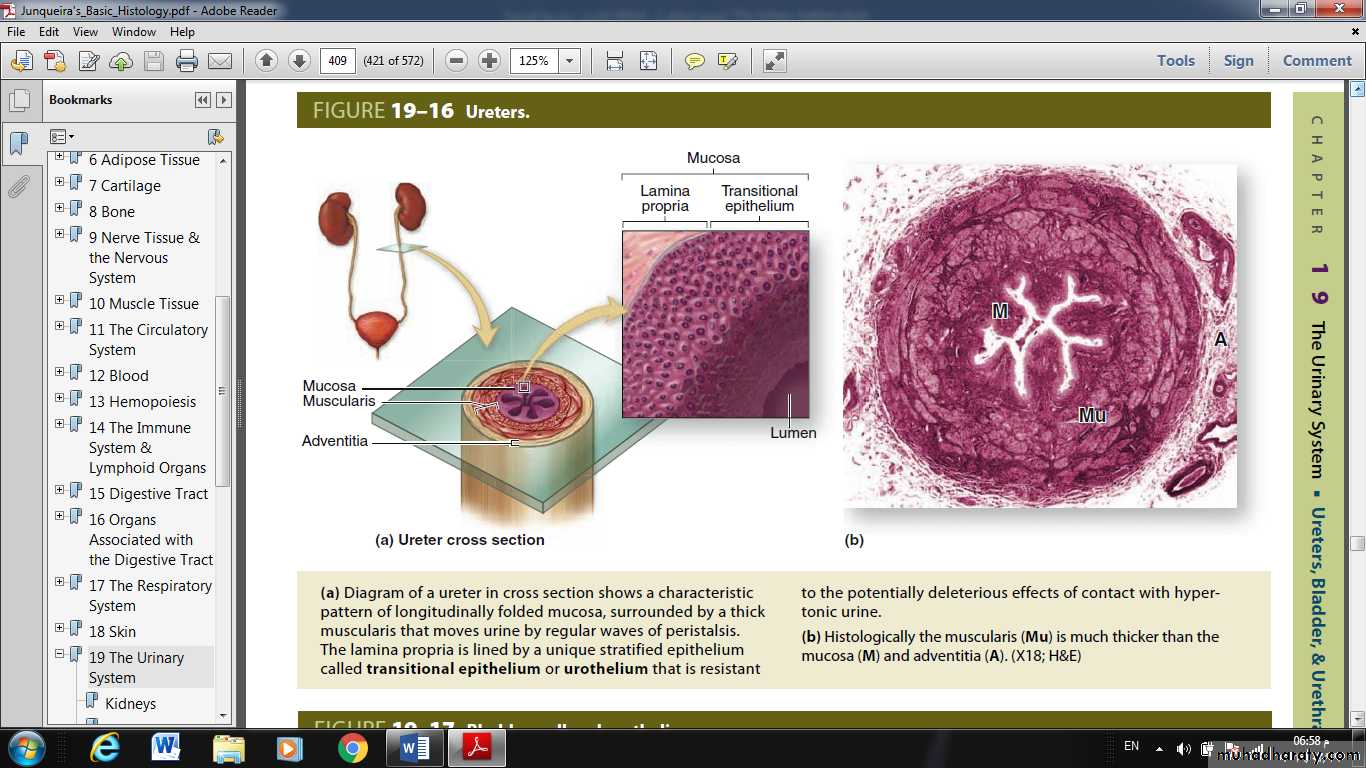
Urine is transported by the ureters from the renal pelvis to the urinary bladder where it is stored until emptying by micturition via the urethra.The walls of the ureters are similar to that of the calyces and renal pelvis, with mucosal, muscular, and adventitial layers and becoming gradually thicker closer to the bladder.
The mucosa of these organs is lined by the uniquely stratified urothelium or transitional epithelium. Cells of this epithelium are organized as three layers:
A single layer of small basal cells resting on a very thin basement membrane;
An intermediate region containing from one to several layers of cuboidal or low columnar cells;
and a superficial layer of large bulbous or elliptical umbrella cells, sometimes binucleated, which are highly differentiated to protect the underlying cells against the potentially cytotoxic effects of hypertonic urine.
The thick muscularis of the ureters moves urine toward the bladder by peristaltic contractions and produces prominent mucosal folds when the lumen is empty.
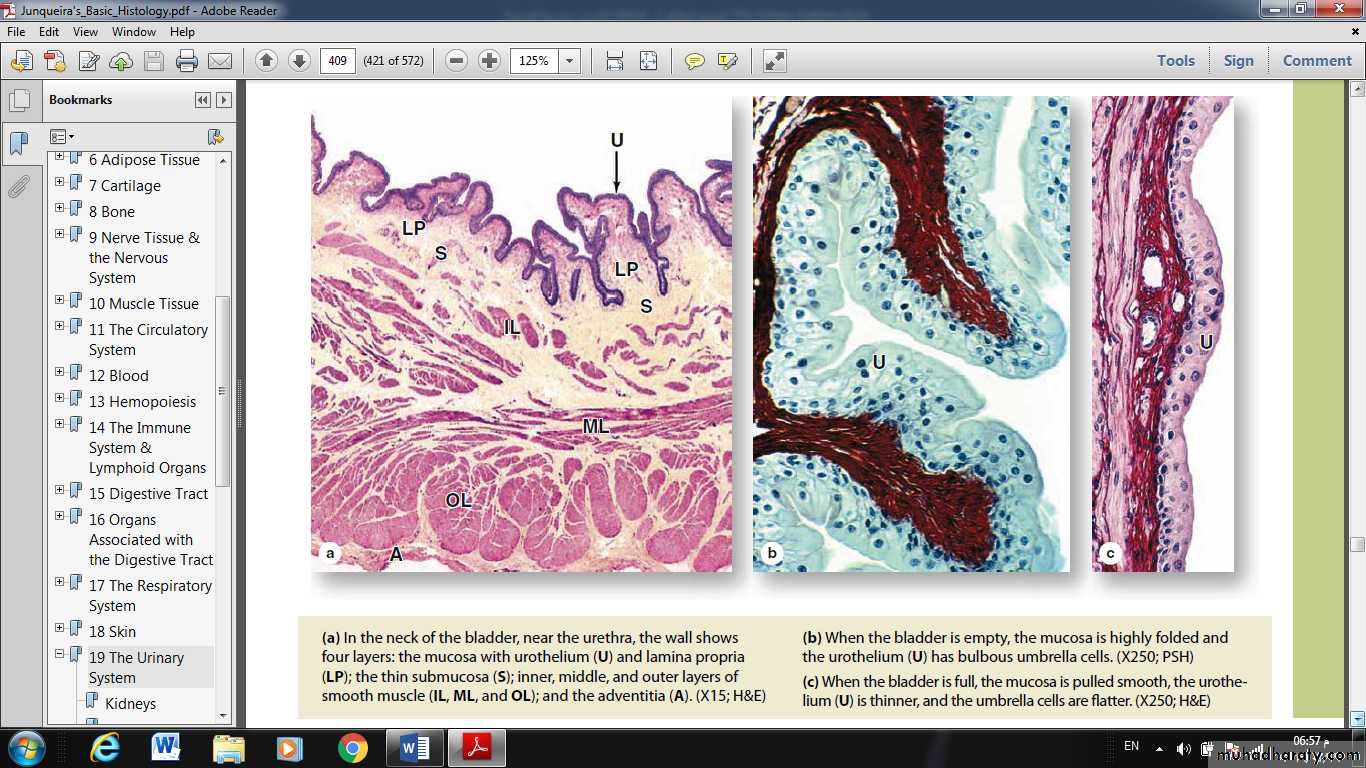
Umbrella cells are especially well developed in the bladder where contact with urine is greatest.
When the bladder is emptied, not only does the mucosa fold extensively, but individual umbrella cells decrease their apical surface area.
The thickness of the full bladder’s urothelium is half that of the empty bladder (5-7 cell layers vs. 2-3 layers),
apparently the result of the intermediate cells being pushed and pulled laterally to accommodate the increased volume of urine.
Urothelium is surrounded by a folded lamina propria and submucosa, followed by a dense sheath of interwoven smooth muscle layers and adventitia.
Urine is moved from the renal pelvises to the bladder by peristaltic contractions of the ureters.
The bladder in an average adult can hold 400-600 mL of urine,
with the urge to empty appearing at about 150-200 mL.The muscularis consists of three poorly delineated layers, collectively called the detrusor muscle, which contract to empty the bladder.
Three muscular layers are seen most distinctly at the neck of the bladder near the urethra.
The ureters pass through the wall of the bladder obliquely, forming a valve that prevents the backflow of urine into the ureters as the bladder fills.
All the urinary passages are covered externally by an adventitial layer, except for the upper part of the bladder that is covered by serous peritoneum.
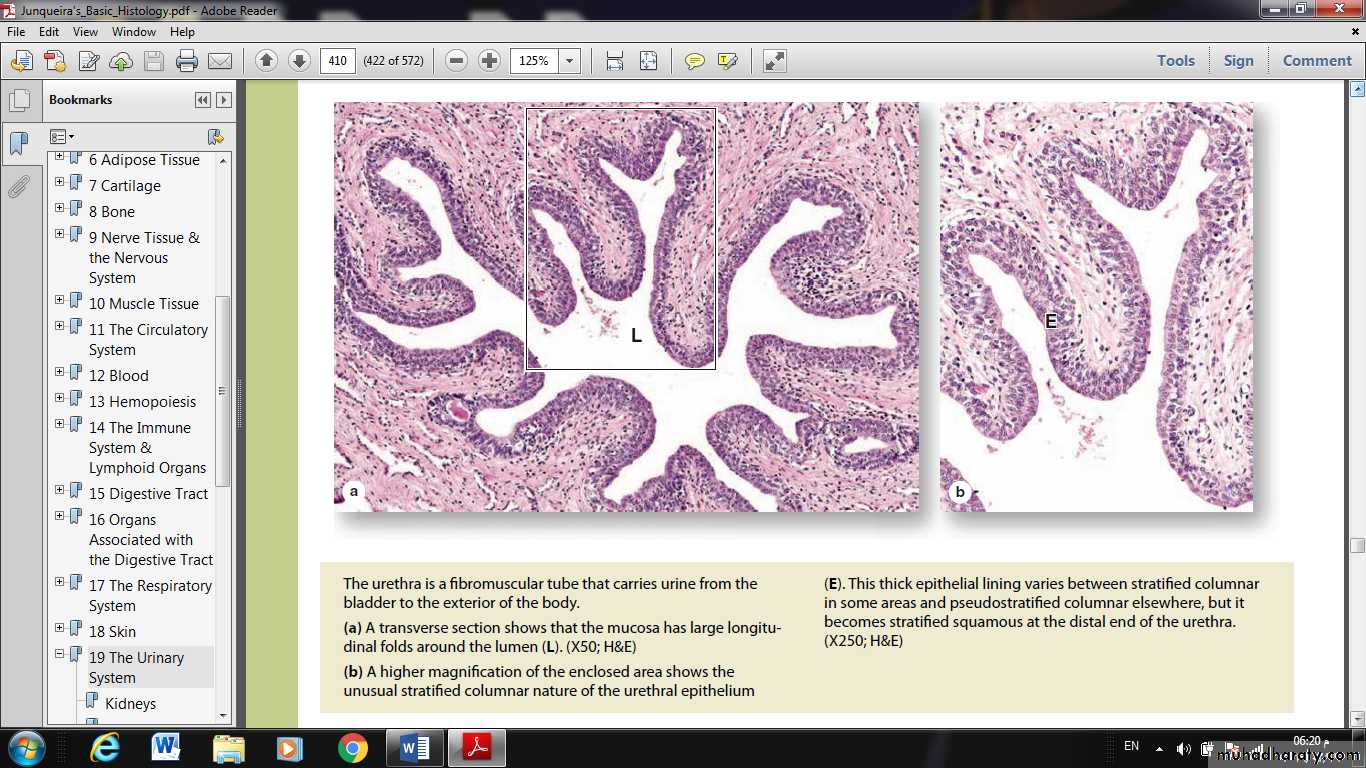
The urethra is a tube that carries the urine from the bladder to the exterior.
The urethral mucosa has prominent longitudinal folds, giving it a distinctive appearance in cross section.The male urethra is longer and consists of three segments:
The prostatic urethra, 3-4 cm long, extends through the prostate gland and is lined by urothelium.
The membranous urethra, a short segment, passes through an external sphincter of striated muscle and is lined by stratified columnar and pseudostratified columnar epithelium.
The spongy urethra, about 15 cm in length, is enclosed within erectile tissue of the penis and is lined by stratified columnar and pseudostratified columnar epithelium, with stratified squamous epithelium distally.
In women, the urethra is exclusively a urinary organ.
The female urethra is a 3- to 5-cm-long tube, lined initially with transitional epithelium which then transitions to nonkeratinized stratified squamous epithelium continuous with that of the skin at the labia minora.The middle part of the urethra in both sexes is surrounded by the external striated muscle sphincter.